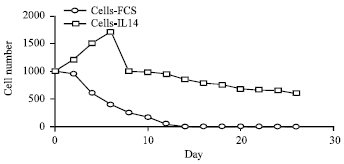
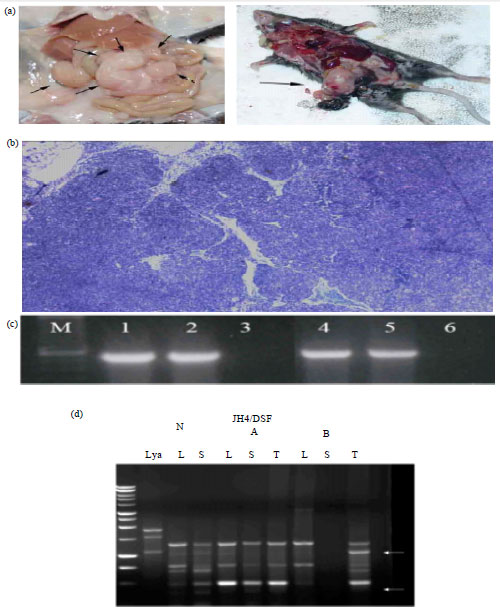
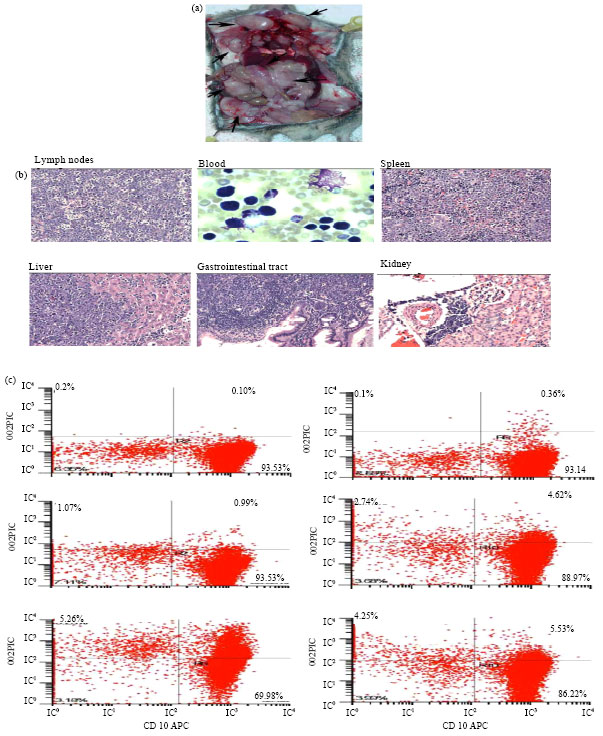
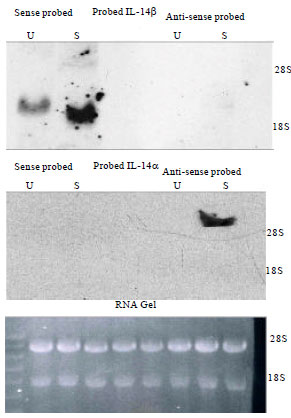
Research Article
IL-14β Transgenic Mice Develop Tumors Consistent with Follicular Lymphoma
Department of Medicine, SUNY at Buffalo School of Medicine and Biomedical Sciences, Buffalo, NY
Long Shen
Department of Medicine, SUNY at Buffalo School of Medicine and Biomedical Sciences, Buffalo, NY
Chongjie Zhang
Department of Immunology, Sichuan University, Sichuan, China
Richard J. Ford
Department of Hemato-pathology, MD Anderson Cancer Center, Houston, Texas
Julian L. Ambrus
Department of Medicine, SUNY at Buffalo School of Medicine and Biomedical Sciences, Buffalo, NY