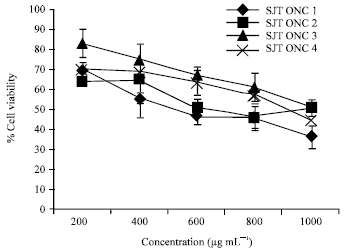
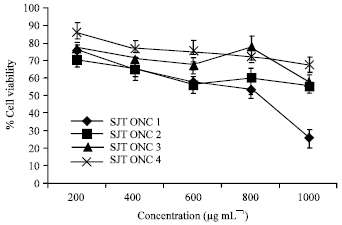
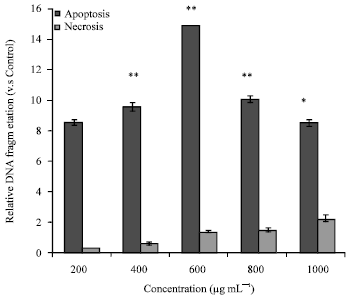
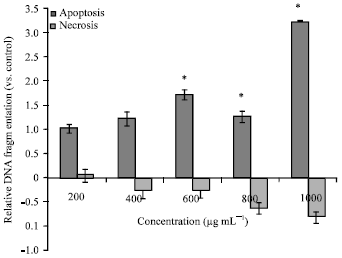
Research Article
Evaluation of Anti Cancer Activity of Polyherbal Formulation
Department of Pharmacology, S.J. Thakkar Pharmacy College, Rajkot, Gujarat, India
H. Dave
Receptor and Growth Factor Laboratory, Gujarat Cancer and Research Institute, Ahmedabad, Gujarat, India
S. Trivedi
Receptor and Growth Factor Laboratory, Gujarat Cancer and Research Institute, Ahmedabad, Gujarat, India
M.A. Rachchh
Department of Pharmacology, S.J. Thakkar Pharmacy College, Rajkot, Gujarat, India
R.H. Gokani
Department of Pharmacology, S.J. Thakkar Pharmacy College, Rajkot, Gujarat, India