Research Article
Genotoxicity Assessment of a Natural Anti-cancer Compound Zerumbone in CHO Cell Lines
Department of Biochemistry and Molecular Biology, Faculty of Medicine and Health Sciences, University of Sana`a, Sana`a, Yemen
Herbal medicine has been gained a worldwide considerable attention which is based on the premise that these herbal plants contain natural substances that can promote health and alleviate diseases. According to World Health Organization, 80% of the earth inhabitants use plant extract or their active compounds in the traditional medicine (Craig, 1999). One fourth of the modern drugs prescribed worldwide are plant derived therapies, where over 121 of these compounds still used in the treatment of various diseases (Rates, 2001).
Zingiber zerumbet Smith (Zingiberaceae), a South East Asian wild ginger and is known as ‘lempoyang’ in Malaysia. It is cultivated in village gardens throughout the tropical and subtropical area and is commonly used in traditional medicine for treatment of a number of illnesses. In some South East Asian traditional medicine, the rhizomes of the plant are usually used as anti-inflammation whiles shoots are used as a condiments. The bioactive compound extracted from Zingiber zerumbet is known as Zerumbone (ZER) that proved to have a colon and lung carcinogenesis suppressive effects (Kim et al., 2009).
The bioactive compound ZER has recently draw the attention of many researchers due to its activity towards many diseases in vitro and in vivo. Its modulatory activity for osteoclastogenesis has been reported recently to be induced in RANKL and breast cancer cells (Sung et al., 2009), as well as a suppressive effects in mouse colon and lung cancinogenesis through multiple modulatory mechanisms (Kim et al., 2009) and colonic tumour marker formation in rats and induces apoptosis in human colorectal cancer cell lines (Murakami et al., 2002). In a dose-dependent manner, the compound was reported to inhibit the growth of human colonic adenocarcinoma cell lines, however, normal human dermal and colon fibroblast were less affected (Murakami et al., 2002, 2004). ZER activity against DES-induced mice Cervical Intraepithelial Neoplasia (CIN) has been reported in our lab (Bustamam et al., 2008). In addition, it has been reported to inhibit both azoxymethane-induced rat aberrant crypt foci and phorbol ester-induced papilloma formation in mouse skin cancer (Tanaka et al., 2001).
Due to the widespread use of herbal remedies as well as modern medicinal products and other household and environmental chemicals, genotoxicity studies are of great interest. Many plant products contain compounds known to cause various diseases or even death in animals and humans as well as synthetic substances present as environmental pollutants and toxicants may cause similar effects (Rates, 2001; Ames and Gold, 1997). Many natural and synthetic compounds have been reported to act as mutagens and/or carcinogens (Ames, 1983; Vargas et al., 1990). A variety of in vitro genotoxicity test systems have been developed including the bacterial mutation assay as the Ames test and cultured mammalian cell systems such as human Peripheral Blood Lymphocytes (PBL) or Chinese Hamster Ovary (CHO) cells, for the screening of potentially mutagenic, carcinogenic and/or teratogenic agents.
This study reports the results obtained on the genotoxic effects of ZER in Chinese Hamster Ovary (CHO) cell lines, by using chromosomal aberrations assay (CAs) and micronucleus (MN) formation as cytogenetic endpoints as well as using the bacterial point mutation assay, Ames test. This paper described the results of those investigations and comments on the in vitro genotoxic effects of ZER in CHO cell line.
The present study was conducted from January 2009 to October 2009 in the University of Putra Malaysia in the Laboratory of Cancer Research MAKNA-UPM.
MTT cytotoxicity assay: ZER (Fig. 1) was extracted in the laboratory of cancer research MAKNA-UPM, University Putra Malaysia, from the rhizomes of Zingiber zerumbet plant (Abdul et al., 2008).
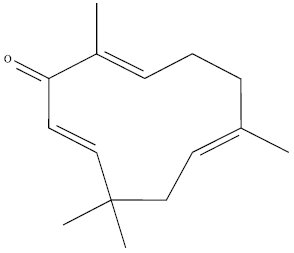 | |
| Fig. 1: | Chemical structure of zerumbone |
Cytotoxicity assay was performed using one fold serial dilution gradient of ZER concentrations, ranging from 2.5-80 μM. The MTT ([CAS 298-93-1] Sigma) assay used has been described by Mossman (1983). All treatments were performed in triplicate and the cells were cultured for 24 h in the presence and absence of ZER. An IC50 is determined from each of the three experiments and the final IC50 was determined by averaging the values of the three experiments (±SD).
Chromosomal aberration assay: It is usual in genotoxicity testing to investigate the potential of a chemical to induce chromosomal damage as well as gene mutations. Therefore ZER was evaluated for its potential to produce chromosome aberrations in CHO. Atypical Chinese Hamster Ovary (CHO) cells (ECACC, UK), contains 21 chromosomes.
Cells were maintained in RPMI 1640 medium (PAA Laboratories GmbH, Germany) supplemented with 10% foetal bovine serum (PAA Laboratories GmbH, Germany). On the day before the experiment, approximately 5x105 cells from a culture with approximately 60-80% confluence were seeded into each T-25 cm2 (Nunc, Denmark) flasks. The cells were incubated overnight in a humidified incubator at 37°C and 5% CO2. The tested compound was prepared as 1 mg mL-1 stock solution in absolute ethanol <1% just prior to use. Mitomycin-C (Sigma, Germany) was used as a positive control. The treatment medium was 5 mL of the cell culture medium with 10% foetal bovine serum and compound concentration range 2.5-80 μM. Cells were cultured in the treatment medium for 24 h.
Analysis of chromosome aberrations: Two hours prior to harvesting, colcemid (PAA Laboratories GmbH, Germany) was added and cells were trypsinized, treated with hypotonic solution and fixed in 3:1 methanol and glacial acetic acid. Finally, the cells were dropped onto chilled slides and stained in 6% Giemsa stain ([CAS 67-56-1] Gibco, Invitrogen, Germany). For each duplicate culture, at least 1000 cells were examined to score mitotic index. Relative Mitotic Index (RMI) was calculated as:
At least 200 metaphase cells from each negative control and ZER-treated culture (200-400 cells per concentration except from the highest concentration only 106 cells were scored) or 150 cells from the positive control were analyzed. The percentages of cells with aberrations from each concentration were compared to the solvent control values using χ2 analyses.
Micronucleus (MN) test: CHO cell cultures were prepared as previously described in the chromosomal aberrations assay. After 22-24 h the media was removed and the cells were washed with PBS or media and fresh media containing 6 μg mL-1 cytochalasin-B (CB [CAS 4930-96-2], Sigma-Aldrich, Germany) was added and incubated for further 22-24 h. One set of negative and positive controls were run for each experimental day.
Harvest, slide preparation and scoring: In this study, the technique utilized to examine binucleated cells for micronuclei in vitro was that described by Fenech (2000). The induction of MN was determined in at least 1000 binucleated cells with the cytoplasm well preserved and clearly surrounded with nuclear membrane, having an area of less than one third of that of the main nucleus (Kirsch-Volders and Fenech, 2001). The criterion for the identification of MN was according to Fenech (1993) and he CBPI was calculated according to OECD guideline number 487 (OECD, 1997) using the following formula:
A minimum of 1000 cells/concentration were analyzed (N).
Ames bacterial mutation assay: Bacterial mutagenicity test was carried out using the Muta-chromoplate™ Kit S-9 version 3.1 (EBPI, Canada) as described by the manufacturer. The mutagenicity test is based on the well-known ‘Ames test’ (Ames et al., 1975) that utilises a mutant strain of Salmonella Typhimurium (TA100) which is defective in the operon gene coding for histidine.
The growing Salmonella Typhimurium TA100 strain culture was added to 96-well flat-bottomed tissue culture plates each containing a fixed concentration (ranging from 5-500 μg mL-1) of ZER dissolved in DMSO (filtered through a 0.22 μm membrane filter) in minimal media. The plates were incubated at 37°C for 5 days in the absence and presence of the S-9 enzyme mixture. Sodium azide for study without S-9 metabolic activation and 2-aminoanthracene for study with S-9 metabolic activation were included as the positive controls in the assay. The scores for the blank plate, the background control plates and the positive control plates obtained were within the manufacturer’s recommendation, indicating the validity of the results obtained for the aqueous extract.
Statistical analysis: Data were analyzed for Chi-square analysis using SPSS version15.0. All statistical tests were performed at the p<0.05 level of significance.
Cytotoxicity analysis: Cytotoxicity assay must be performed first on the chemicals that are not known to cause any chromosome aberrations for the evidence of cytotoxicity in the system chosen for genotoxicity testing. This would help the establishment of a Maximum Tolerated Dose (MTD) of the chemical being tested. In this study, the MTT cytotoxicity assay was employed in determining the dose response of ZER. The IC50 value obtained for ZER was shown to be 20.8 (±5.1) μM (Fig. 2).
Mitotic index analysis: A twofold series of six concentrations of ZER was determined based on the IC50 obtained from the MTT cytotoxicity assay. The Relative Mitotic Index (RMI) values (Table 1) revealed an increasing inhibitory effect with increasing concentrations of ZER for the three lower concentrations while the inhibitory effect restored for the higher concentrations. On the other hand, mitotic index for the lower test concentration was found not to be significantly different from that seen in the untreated control (p>0.05, Table 1) while the next three concentrations were found to significantly reduce the mitotic index (p<0.001). In contrast, the mitotic index of the two highest concentrations was observed to be non-significantly reduced compared to the control.
 | |
| Fig. 2: | Dose response curve of zerumbone against CHO cell lines |
| Table 1: | Mitotic index and relative mitotic index (RMI) observed for CHO cultures treated with different concentrations of ZER and controls |
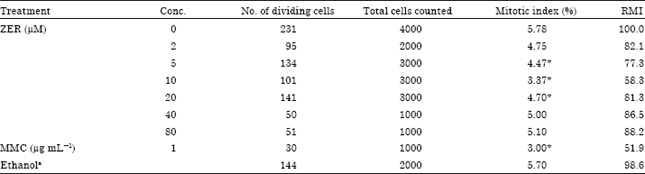 | |
| aEthanol in a final concentration of <1.0%, * Significantly different from the control at p<0.05 | |
Chromosome aberrations (CA) assay: The CA assay, clastogenicity determined was not significantly observed in the untreated control or the solvent treated control. Positive control (MMC) was significantly shown to induce chromosome aberrations. On the other hand, all the clastogenic indices of ZER were found to induce chromosome aberrations in an increasing dose response pattern (Table 2). Gaps, breaks as well as interchanges, endoreduplications, ring chromosomes and dicentrics were the main types of aberration induced by ZER. In treated cultures of CHO cell line the numbers of breaks were found to be significantly and dose-dependently increased (p<0.01). However, ring chromosomes and dicentrics were observed to be significantly increased in all treated cultures except the lowest and highest treatment concentrations, whereas endoreduplications were observed to be significantly increased in the two highest concentrations (p<0.05). The number of aberrant cells with structural aberrations including or excluding gaps recorded in cultures treated with the different drug concentrations increased significantly p<0.01-0.001 (Table 2) when compared with the untreated groups. Fisher’s exact test was used to test the statistical significance of the CA results.
MN induction: Treatment concentrations used in MN test were 5.0-80.0 μM ZER. The potential of ZER to induce micronucleus is shown in Table 3. Treatment of CHO cells with ZER caused dose-dependent MN induction.
| Table 2: | Sum of the frequencies of chromosome aberrations induced in CHO cultures treated with different concentrations of ZER and the positive and negative controls |
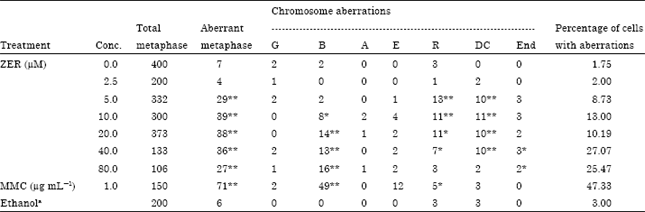 | |
| G: Gap, B: Breaks, A: Acentric, E: Chromatid exchange, R: Chromosome ring, DC: Dicentric, END: Endoreduplication. aEthanol in a final concentration of <1.0%. * Significantly different from the control at p<0.05. ** Significantly different from the control at p<0.001 | |
| Table 3: | Frequencies of micronucleus (MN) formation and cell cycle kinetics on CHO cultures treated with different concentrations of ZER together with Mitomycin C treated and untreated cultures |
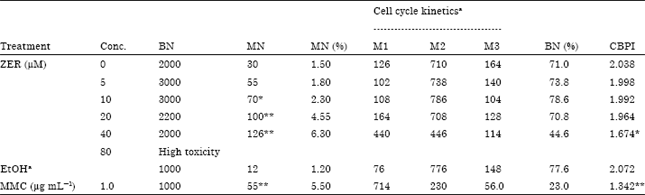 | |
| BN: Binucleate, MN: Micronucleus, aThe No. of mononucleated (M1), binucleated (M2) and polynucleated (M3) cell per 1000 cells were quantitated for cell cycle kinetic analysis. * Significantly different from control p<0.05.** Significantly different from control p<0.001 | |
An increased number of binucleated cells with micronuclei was found at the higher concentrations (>5.0 μM) of ZER and was statistically significantly different from the control. In the cell cycle kinetics analysis or CBPI, treatment with ZER in concentrations more than 20.0 μM was found to exert an inhibition of cell proliferation in CHO cells (Table 3).
Mutagenicity of ZER in Salmonella Typhimurium TA100 strain: The assay was carried out in vitro using one histidine-requiring strain of Salmonella Typhimurium, strain TA100 with and without a metabolic activating enzyme (S9). The bacterial strain was treated with ZER at 5, 50 and 500 μg mL-1, respectively. ZER mutagenicity at various concentrations tested was determined from the statistical table provided by the manufacturer. According to the manufacturer table, the level of mutagenicity was classified as strongly mutagenic if p<0.001, moderately mutagenic if p<0.01 and mildly mutagenic if p<0.05. The results obtained in the presence and absence of S-9 metabolic activation are presented in Table 4.
| Table 4: | No. of positive wells in the background control plates, the positive control plates and the plates treated with various concentrations of ZER in the presence and absence of S9 metabolic activation |
 | |
| !: Absent, +: Present | |
All the tested concentrations of ZER (5-500 μg mL-1) were found to be not mutagenic in the absence and the presence of S-9 metabolic activation as the numbers of positive wells in the treated plates were not statistically different from the results obtained in the background plate, in contrast at high ZER levels the number of positive wells were reduced compared to the background. The positive controls, sodium azide and 2-aminoanthracene, were strongly mutagenic as the number of positive wells in the positive control plates were significantly higher than that in the background plate (p<0.001). However, the number of positive wells in the highest concentration treatment was less than that in the background plate.
Genotoxic agents have the potential to interact with DNA and may cause DNA damage. CA occurs in proliferating cells and is regarded as a manifestation of damage to the genome. CA assay has been commonly used as a test of mutagenicity in order to evaluate cytogenetic responses to chemical exposure. In the present study, ZER significantly decreased the mitotic index in cultures of Chinese hamster ovary cell line. The concentration range of 5.0-20.0 μM was observed to decrease the mitotic index at significant level of p<0.01 in contrast to the lower and higher concentration levels which found not to significantly reduce the mitotic index.
The genotoxic effect of the test compound was tested at concentrations started from 2.5-80 μM. These tested concentrations were chosen according to the cytotoxicity test. The results of the present study demonstrated that the frequency of breaks, gaps, fragments, exchanges, dicentrics and endoreduplications were increased significantly in cultures of Chinese hamster cell line treated with ZER when compared with the control. The number of total aberrant cells recorded at concentrations of 5.0-80.0 μM were significantly increased at p<0.001 when compared with untreated control. The percentage of structurally damaged cells in the MMC (positive control) treatment group was statistically increased compared to the solvent control indicating the responsiveness of the cells in this test system.
The presence of dicentric and ring chromosomes may suggest telomeric loss or telomeric associations as well as chromatid/chromosome breaks. Telomeric loss starts chromosomal breakage-fusion-bridge cycle which contains DNA double strand break near a telomere, sister chromatid fusion and bridge formation in the mitosis (anaphase) and results in the production of mitotically unstable chromosome aberration, i.e., ring chromosome, dicentric chromosome or telomeric association in metaphase cells (Gisselsson et al., 2000; Lo et al., 2002). Endoreduplication in eukaryotes is a process that involves DNA amplification without corresponding cell division (Sumner, 1998; Sugimoto-Shirasu et al., 2002). Regardless of the molecular mechanism, inhibition of DNA topoisomerase II results in the formation of endoreduplication (Sumner, 1998; Pastor et al., 2002; Cortes and Pastor, 2003). An increase in the number of cells with endoreduplicated chromosomes may indicate that the test substance has the potential to inhibit cell cycle progression. It has been demonstrated that ZER inhibits the proliferation of leukemia cells via the induction of G2/M cell cycle arrest followed by apoptosis (Xian et al., 2007) supporting the hypothesis that ZER halt the cells in the G2/M causing the formation of diplochromosomes (endocycle).
MN assay is a widely used cytogenetic method to assess in vitro chromosomal damage. Analysis of the frequency of occurrence of micronuclei in treated cells provides a comparatively rapid and sensitive indication of both chromosomal aberrations and chromosome loss that lead to numerical chromosomal anomalies (Azeddine et al., 1998; Matsuoka et al., 1999). Micronuclei are chromatin masses in the cytoplasm with the appearance of small nuclei that arise from chromosome fragments at anaphase or from acentric chromosomal fragments. They provide a quantifiable measure of recent DNA injury that result from when acentric fragments or whole chromosomes are left behind the main nucleus at telophase (Matsushima et al., 1999). An increase in the percentage of MN in a population of cells indicates chromosomal damage occurred as a result of an exposure to either clastogenic or an aneuploidogenic effect (Kirsch-Volders et al., 2003). Present study showed that exposure of CHO cells to ZER significantly and dose-dependently increased the frequency of MN. These results of micronucleus formation support our observations that ZER causes chromosomal damage in Chinese hamster cell lines, (Al-Zubairi et al., 2010a) indicating its potential to cause genotoxic effects. In contrast to the results of the mutagenicity Ames assay, in the results were found to be negative, inducating ZER to have no potential to cause point mutations in the Salmonella Typhimurium strain TA100. In addition, data on the effects of ZER on normal human peripheral blood lymphocyte chromosomes showed to have less impact when compared to the commercial anticancer drug cisplatin, as well as the higher concentrations were found to exert less significant effects on the chromosomes (Al-Zubairi et al., 2010b).
Structurally, ZER carried no obvious structural alerts and no obvious mechanism-based genotoxicity had been identified (e.g., nucleoside analogs, topoisomerase or inhibitors) that can cause a genotoxic effects, instead these effects were found to be significant in the present investigation. This unexplained genotoxicity is much more common in the in vitro cytogenetics (Snyder et al., 2004, 2006). Some of these positive responses may be due to true covalent adducts formation whereas, others are most likely cytotoxicity artefacts (Galloway, 2000) and still others may be due to non-covalent drug/DNA interactions, i.e. DNA intercalation or groove-binding (Snyder et al., 2006; Snyder, 1998).
The present results provide evidence that ZER compound in high concentrations have a genotoxic and cytotoxic effects on cultured Chinese hamster cell line, bringing into highlight the need for further studies to better understand the molecular mechanisms of action of ZER compound for a better comprehension. Thus, results found, at least under the experimental conditions used in the present study, showed the ability of this compound to induce genotoxicity and cytotoxicity in vitro in CHO cells while it failed to induce mutagenic effects on Salmonella Typhimurium strain TA100.
The author would like to extent his utmost gratitude and appreciation to MOSTI (Ministry of Science, Technology and Innovation) and The National Cancer Council Malaysia (MAKNA) for providing the research grants IRPA No: 06-02-04-0720-EA001. The author also would like to convey his thanks to Dr Ahmad Bustamam, for his supervision and Dr Syam Mohan, Dr Siddig I Abdelwahab for their help in completing this work as well as Mr Will Lush from EBPI, Canada, for his help in duplicating the Ames test.