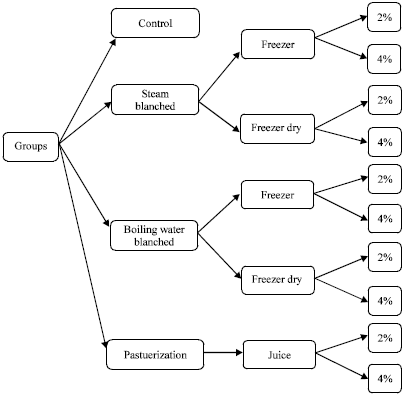
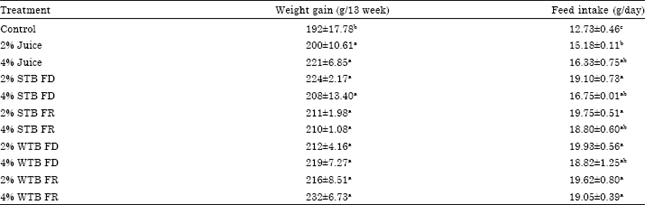
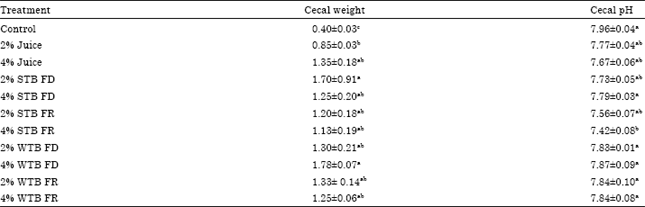
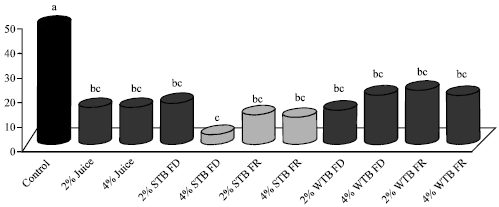
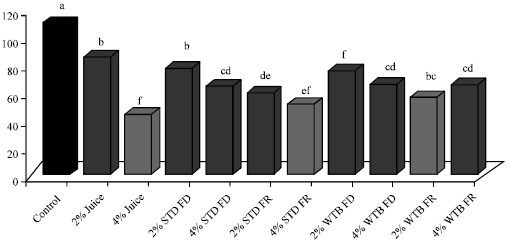
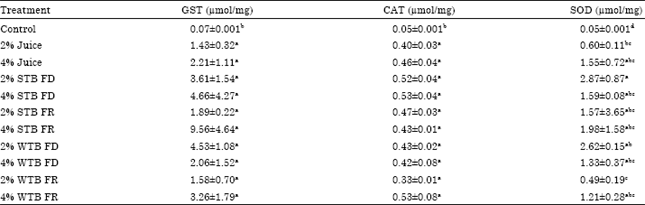
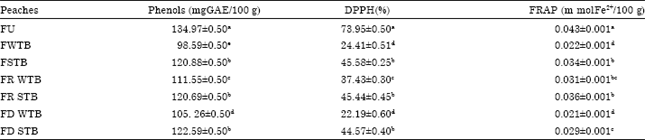
Research Article
Effects of Processed Peaches (Prunus persica) in Reducing Azoxymethane (AOM) Induced Aberrant Crypt Foci (ACF) in Fisher 344 Male Rats
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA
J. Boateng
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA
L. Shackelford
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA
S. Appiah
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA
K. Campbell L. T. Walker
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA
M. Verghese
Nutritional Biochemistry Laboratory, Department of Food and Animal Sciences, Alabama A and M University, P.O. Box 1628, Normal, AL 35762, USA