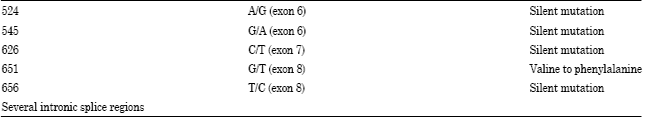Review Article
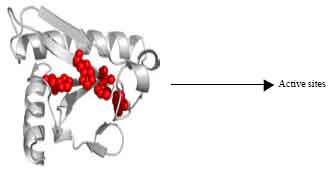
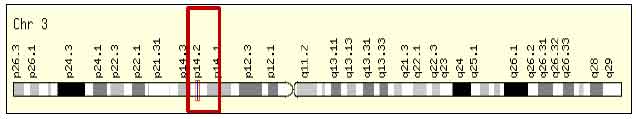
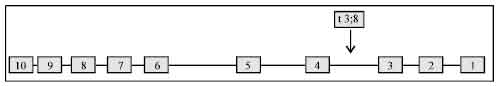
Introduction to the Role of Fragile Histidine Triad (fhit) Gene in Cancer: A Review of Literature with Special Emphasis on Cervical Carcinoma
Golden Homes, Tolichowki, Hyderabad, Andhra Pradesh, India
S. Kiranmayee
Stapenhill Medical Centre, Burton-on-Trent, Staffordshire, DE15 9QD, United Kingdom
V.K. Bammidi
Stapenhill Medical Centre, Burton-on-Trent, Staffordshire, DE15 9QD, United Kingdom
A.P. Shaik
Jawaharlal Nehru Institute of Advanced Studies, Buddha Bhavan, MG Road, Secunderabad, Andhra Pradesh, India
K. Jamil
Jawaharlal Nehru Institute of Advanced Studies, Buddha Bhavan, MG Road, Secunderabad, Andhra Pradesh, India