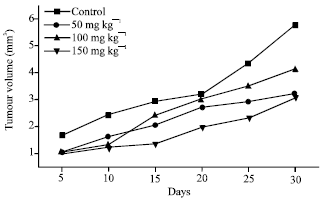
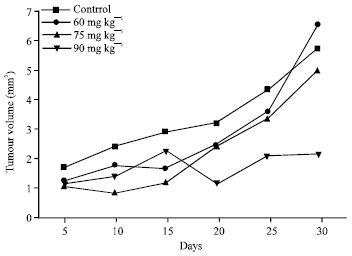
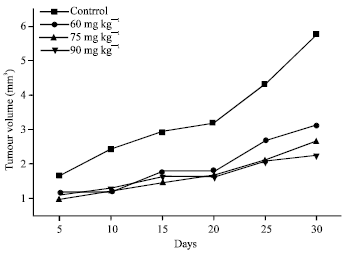
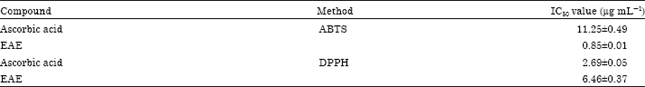
Research Article
Antitumor Activity of Hexane and Ethyl Acetate Extracts of Tragia involucrata
Department of Biochemistry, Mangalore University, Cauvery Campus, Madikeri-571 201, Karnataka, India
M. Gopal
Department of Biochemistry, Davanagere University, Shivagangothri, Davanagere-577 001, Karnataka, India
N. Suchetha Kumari
Department of Biochemistry, Nitte University, Mangalore-575018, Kanrnataka, India