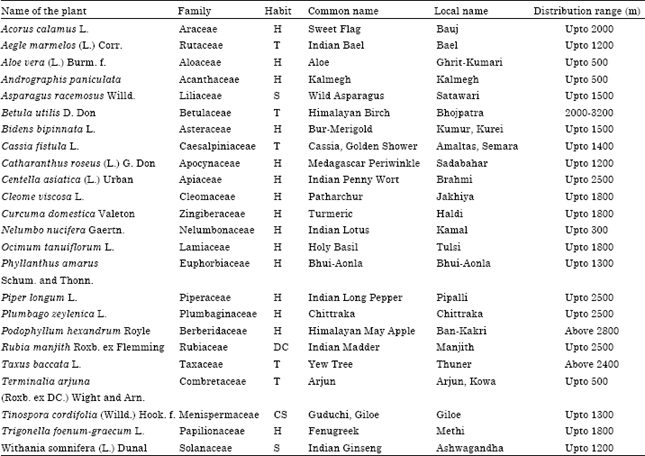
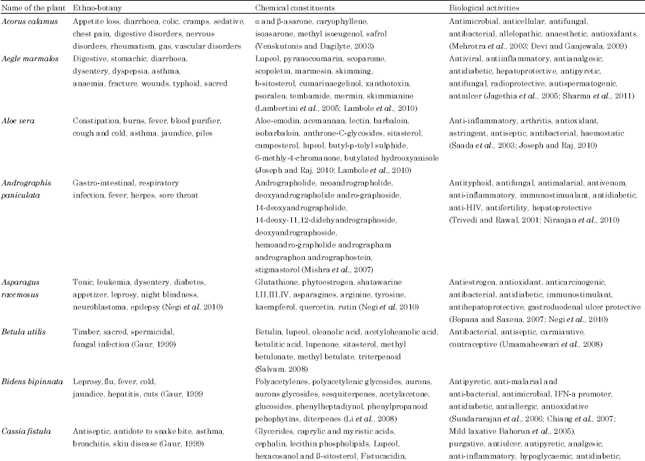
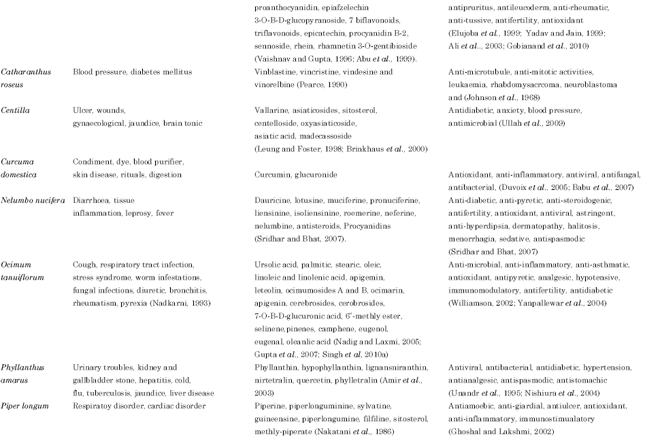
Review Article
Anti-cancerous Plants of Uttarakhand Himalaya: A Review
Herbal Research and Development Institute, Mandal, Gopeshwar-246 401, Chamoli, Uttarakhand, India
J.S. Negi
Herbal Research and Development Institute, Mandal, Gopeshwar-246 401, Chamoli, Uttarakhand, India
A.K. Bhandari
Herbal Research and Development Institute, Mandal, Gopeshwar-246 401, Chamoli, Uttarakhand, India
R.C. Sundriyal
Herbal Research and Development Institute, Mandal, Gopeshwar-246 401, Chamoli, Uttarakhand, India