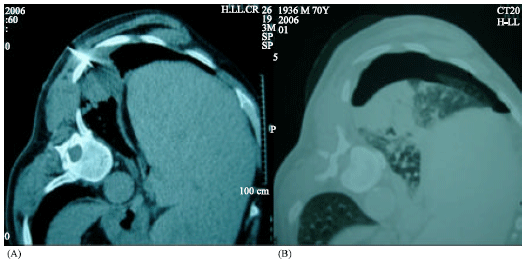
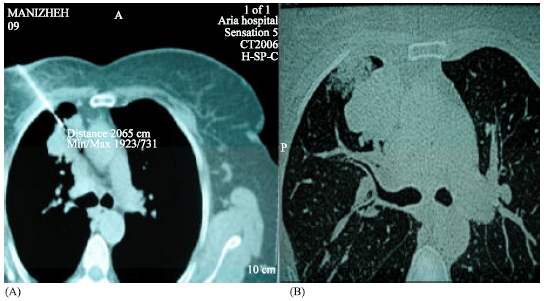
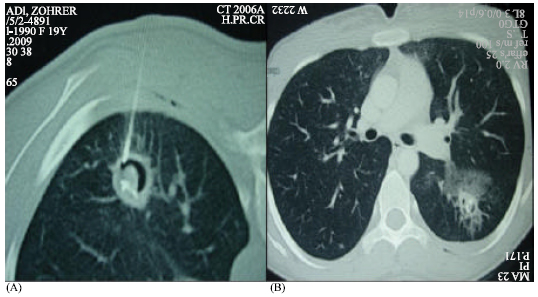
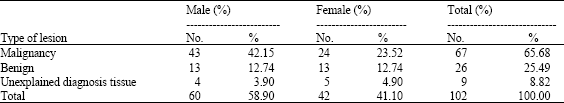
Research Article
Study the Complications of Ct-guidance Fine Needle Biopsy in Intra-thoracic Masses
Department of Radiology, Imam Khomeini Hospital, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
M.S.Z. Sharifi
Radiology Residents, Imam Khomeini Hospital, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
F. Rahim
Physiology Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran