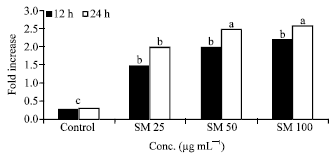
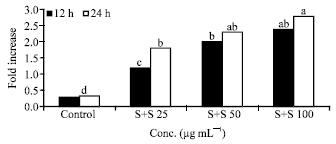
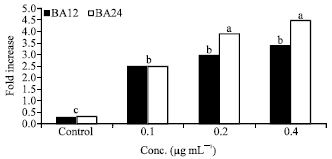
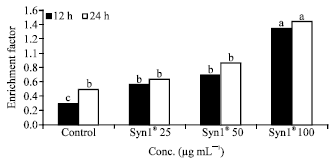
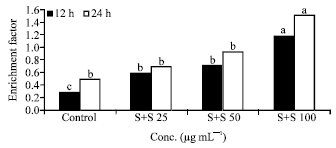
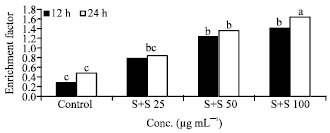
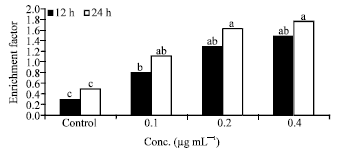
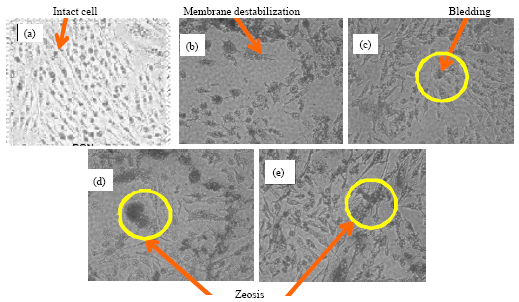
Research Article
Anticancer Effects of Prebiotics Synergy1® and Soybean Extracts: Possible Synergistic Mechanisms in Caco-2 Cells
Nutritional Carcinogenesis Laboratory, Department of Food and Animal Sciences, Alabama A and M University, Normal, AL, USA
M. Verghese
Nutritional Carcinogenesis Laboratory, Department of Food and Animal Sciences, Alabama A and M University, Normal, AL, USA
J. Boateng
Nutritional Carcinogenesis Laboratory, Department of Food and Animal Sciences, Alabama A and M University, Normal, AL, USA