Research Article
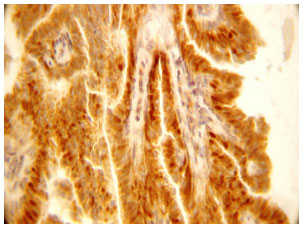
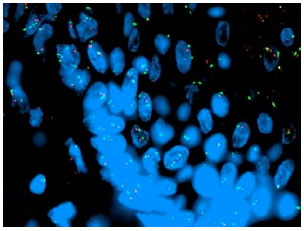
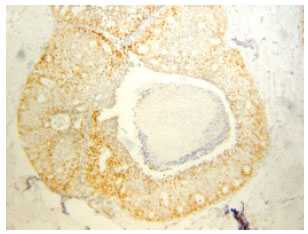
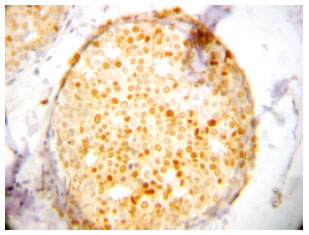
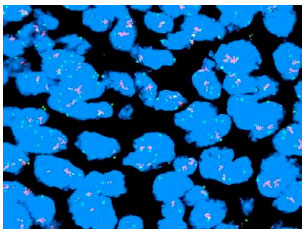
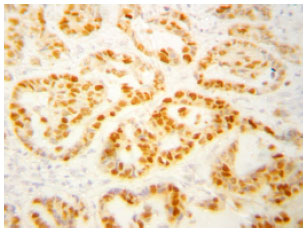
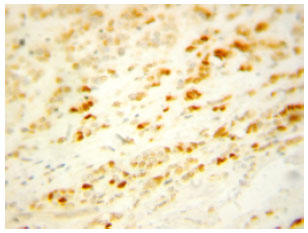
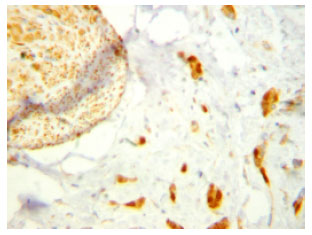
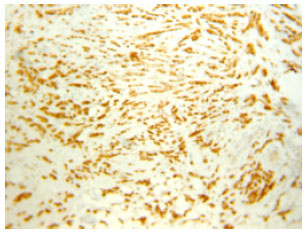
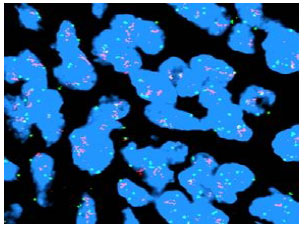
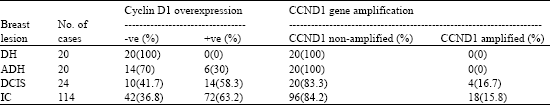
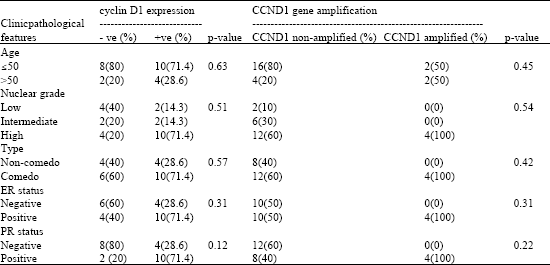
Significance of Cyclin D1 Overexpression and Amplification in Ductal Hyperplasia, Carcinoma in situ and Invasive Carcinoma in Egyptian Female Breast
Department of Pathology,
Moatasem M. Aly
Department of Surgery, Faculty of Medicine, Minia University, Egypt