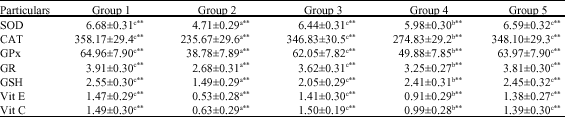
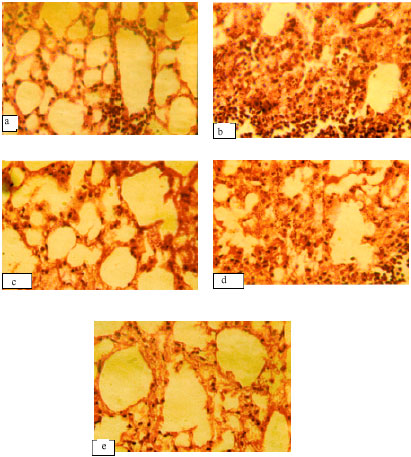
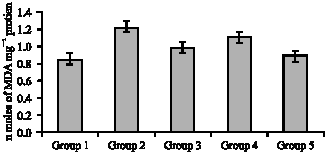Research Article
Protective Effect of Flavanoidal Fraction of Indigofera tinctoria on Benzo (α) Pyrene Induced Lung Carcinogenicity in Swiss Albino Mice
Department of Zoology, Unit of Biomonitoring and Management, University of Madras, Guindy Campus, Chennai-600025, India
Ravichandran Ramanibai
Department of Zoology, Unit of Biomonitoring and Management, University of Madras, Guindy Campus, Chennai-600025, India