Research Article
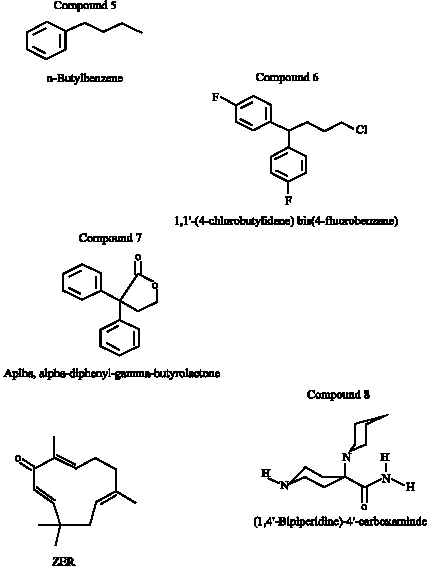
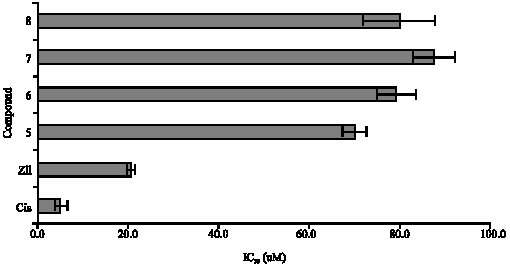
Anti-Tumor Activities of Analogues Derived from the Bioactive Compound of Zingiber zerumbet
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia
A.B. Abdul
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia
H.C. Yee1
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia
A.S. Alzubairi
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia
M.M. Elhassan
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia
M.M. Syam
UPM-MAKNA Cancer Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Serdang, 43400, Selangor, Malaysia