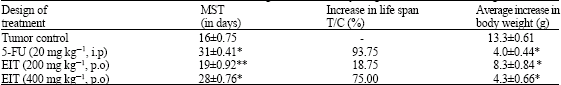
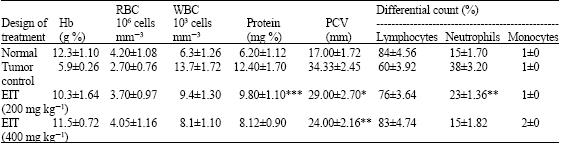
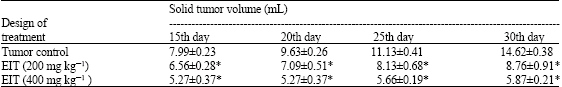
Research Article
Antitumour Activity of Indigofera trita on Ehrlich Ascites Carcinoma Induced Mice
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Vinayaka Missions University, Salem, 636 008, Tamilnadu, India
Balasundaram Jayakar
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Vinayaka Missions University, Salem, 636 008, Tamilnadu, India
Balasubramanian Rajkapoor
Department of Pharmacology, Vel's College of Pharmacy, Velan Nagar, Pallavaram, Chennai, 600 117, Tamilnadu, India