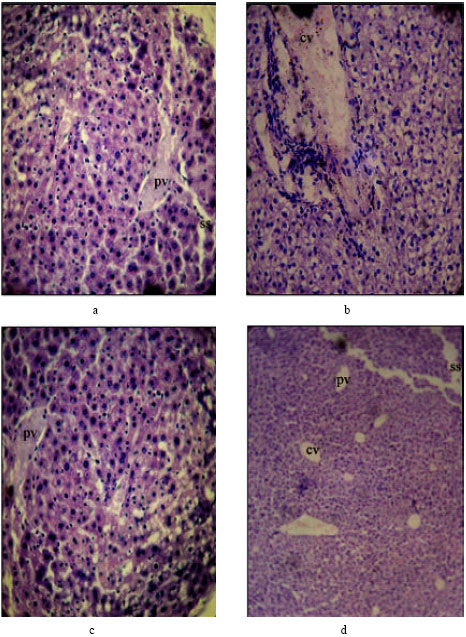
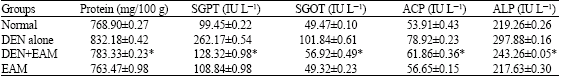Research Article
DEN-Induced Cancer and its Alleviation by Anisomeles malabarica (L.) R.Br. Ethanolic Leaf Extract in Male Albino Mice
Plant Tissue Culture Unit, Department of Plant Biology and Plant Biotechnology, St. Joseph's College (Autonomous), Tiruchirappalli-620 002, India
A. Mahesh
Plant Tissue Culture Unit, Department of Plant Biology and Plant Biotechnology, St. Joseph's College (Autonomous), Tiruchirappalli-620 002, India
L. Cindrella
Computational Chemistry Unit, Department of Chemistry, National Institute of Technology, Tiruchirappalli-620 015, India