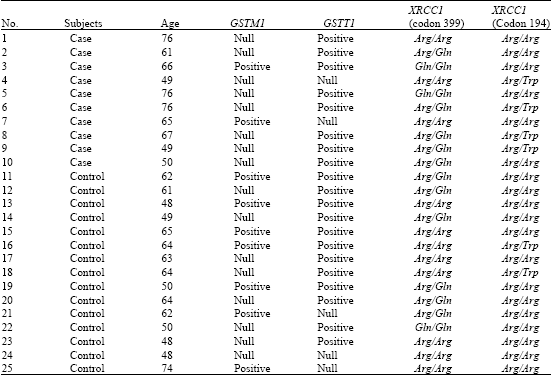
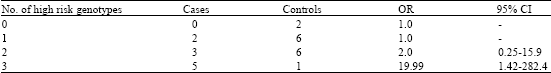Research Article
Combination of XRCC1, GSTM1 and GSTT1 Genetic Polymorphisms and Susceptibility to Male Breast Cancer
Department of Biology, Khatam University, Tehran, Iran
Shahpour Omidvari
Department of Chemotherapy, Shiraz University of Medical Sciences, Shiraz, Iran
Mostafa Saadat
Department of Biology, College of Sciences, Shiraz University, Shiraz 71454, Iran