ABSTRACT
Participants in the PLCO-Hawaii Cancer Screening Trial were given a self-administered 36-item health status questionnaire. This questionnaire includes eight health concepts relevant to the quality of life and self-perceptions of health. These are converted into two summary scales, self-perceived physical health status and self-perceived mental health status. The initial hypothesis of a negative effect of receiving an abnormal cancer-screening outcome to one’s Health Related Quality of Life is not supported for either self-perceived physical health status or for self-perceived mental health status. Receiving notification of a cancer diagnosis does not produce an additional negative effect. There is no difference in health related quality of life between those in the screened group and controls who were not screened at any of the three time periods, baseline, first follow-up or second follow-up. There was no difference in health related quality of life between genders, age groups or ethnicities, except that in the screened group there was a reduction in the physical summary score for the oldest age group between base line and first follow-up assessments.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ijcr.2006.277.289
URL: https://scialert.net/abstract/?doi=ijcr.2006.277.289
INTRODUCTION
Background
Much research has been concerned with identifying risk factors and the characteristics of those at risk for various chronic diseases. One such study is the Behavioral Risk Factors Surveillance Survey conducted nationwide since the 1980’s (BRFSS: http://www.cdc.gov.brfss/). While there has been considerable progress in developing screening techniques, much less attention has been paid to the patient’s perspective and response to the process of disease screening and diagnosis.
This study was originally proposed in the early 1990’s when research on cancer screening outcomes and associated effects on HRQL was in its infancy. Health related quality of life (HRQL) has become increasingly studied with respect to various chronic diseases (Manocchia et al., 1998). We have, in the past, ignored the importance of individual characteristics such as ethnic background and culture, as well as other social-cultural factors in understanding the utility of screening, detection and treatment of diseases like cancer. We know, regardless of various other factors, culturally and ethnically defined quality-of-life has a significant impact on both the process of maintaining health and in dealing with disease or illness (Stafford, 1978). This study takes into account various demographic characteristics along with various aspects of self-perceived health related quality of life, in helping to better understand the psycho-social-cultural effects of disease screening. These dimensions of HRQL are addressed in the survey instrument (SF-36) that was developed out of the Medical Outcomes Study (MOS).
Over half of the Medical Outcomes Study (MOS) population studied in developing the SF-36 was over 60 years of age, about 40% were eligible for Medicare and all had one or more chronic conditions (Ware, 1993). This profile is similar to that of the PLCO population. The content of the SF-36 items reflect common themes found in health insurance literature, thus allowing for comparison with findings of other studies. Rather than focus particularly on instruments designed for patients diagnosed with cancer (Spitzer et al., 1981), the SF-36 allows the researchers to measure eight different, yet interrelated aspects of health. The SF-36 has been used in hundreds of clinical trials. It represents a generation of psychometric instruments that are short, self-administered and comprehensive (Manocchia et al., 1998; Ware, 1997).
The SF-36 has also been utilized in other studies at the Pacific Health Research Institute (PHRI), where it was shown that ethnic differences were an important aspect of health maintenance and disease screening. The Hawaii PLCO participant population is ethnically diverse. This diversity is reflected in the culturally influenced attitudes, knowledge and beliefs about cancer contained in the participant’s responses to the survey. In some cultures, they would rather not know if they have an incurable cancer (Perez-Stable et al., 1992). Such beliefs may delay or prevent many from seeking medical care (Vernon et al., 1985). This may be true among some of Hawaii’s cultural groups.
Participants in the PLCO-Hawaii screening trial were given a self-administered 36-item health status questionnaire. This questionnaire includes eight health concepts relevant to the quality of life and self-perceptions of health that may have affected participant experience with the PLCO screening trial.
The particular advantage of this study is that participants were closely monitored for health changes during the entire follow-up period of the study so that patterns of responses can be accounted for. The original study with a baseline and one-year follow-up was extended an additional three years for a total follow-up period of four years. Thus, we have both a short follow-up period and a long follow-up period in order to test the cumulative effect of multiple cancer screenings over time. Such tests have been shown to be very sensitive (Katz et al., 1992).
Study Objectives and Hypotheses
The Overall Goals of This Ancillary Project were
| • | Quantify and track the self-perceived health status of screening and control participants in the Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial (PLCO) at the Hawaii site. |
| • | Evaluate the participant’s attitudes toward their own health, including the impact of their cancer screening experience. |
This will provide insight into the patterns of health perceptions relevant to improved health care. The overall objectives of the PLCO Cancer Screening Trial is to determine if periodic screening of men and women age 55-74 (at entry) for cancers of the prostate, lung, colorectum and ovaries will reduce the mortality from the targeted cancers as compared to a control group receiving usual health care. This study provides the psychometric measures needed to assess participant’s health perceptions through the screening and during their first years of follow-up.
The specific study objectives of this ancillary study were:
| • | Measure the effect of screening for cancer when an abnormal screen is obtained. An abnormal screening result is expected to reduce the participant’s perception of health status (health related quality of life). |
| • | Measure the effect of screening regardless of outcome and compare baseline (BL), prior to the PLCO screening, with the 1st year follow-up (1F/U), after the 1st PLCO screening results are known and 4th year PLCO follow-up (2F/U), after the 4th year PLCO screening results are known. |
| • | Compare respondents in the screened group with controls regarding self-reported health related quality of life at BL, 1F/U and 2F/U. |
| • | Determine the effect of gender, age and ethnicity on self-reported health related quality of life (HRQL). |
This study addresses each of these objectives. It was not intended to address the issue of the effect of false positive cancer screenings on HRQL. This study fits within the National Cancer Institute (NCI) framework for cost-effectiveness and HRQL measures. The PLCO participants randomized in Hawaii from April 1997 through September 1997 were selected as study participants.
Definitions and Survey Procedures
The main construct of this study has to do with the participants in the screened group being told the results of their cancer screening tests. The controls received no screening tests from the study. The screened group participants received annual follow-up screenings, which over time increased their likelihood of having an abnormal (positive) screening result during the four-year follow-up period. The critical factor is that over time (i.e., Baseline, 1st follow-up (after 1 year) and 2nd follow-up (after 3.5 to 4 years) they could have been screened abnormal on one or more tests at each or any of those times. Most of these participants had no abnormal screens at any of the three times and the rest of those who did have abnormal screens did so at one or more of the times. The data was collected so that at follow-up it was determined whether the participant had an abnormal screening event or not. The protocol is as follows:
PLCO participants were randomly assigned as to the screened group and control group and were followed over time, with those in the screened group receiving screenings for cancer (prostate, lung and colorectal for males and lung, colorectal and ovarian for females). Some of the screenings were done less often than annually, i.e., colorectal. However, all in the screened group received one or more cancer screenings at each annual follow-up. If any of the screenings at a given follow-up tested positive, the participant was considered to have an abnormal screening result at that follow-up. At baseline and at the first follow-up (1F/U) there was just one opportunity to receive one or more abnormal screens. By the second follow-up (the last study inquiry) three years had passed since the first follow-up and during that time the participants in the screened group had been screened at least three times. Even though PLCO staff continued to encourage participants with abnormal screens to obtain independent verification (re diagnostic tests with an outside physician), not all did. Thus, even though not all of the abnormal screens reported in this study have been independently verified by the time of the data analysis, a large majority of them have since been verified.
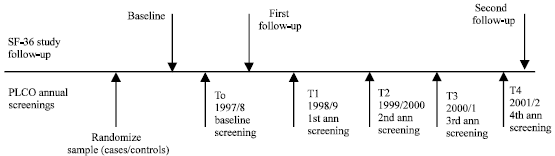 | |
| Fig. 1: | Timeline/flowchart of SF-36 ancillary study follow-up and PLCO annual screenings Note: SF-36 Study participants include a cohort of PLCO participants selected in 1997 Source: PLCO-Hawaii SF-36 Ancillary Study |
Also, there is no attempt to measure the occurrence of false positive screenings, due to the above reason. The immediate effect of a false positive screening result is posited to be the same as a true result, only after the participant learns that the screening result was incorrect would it effect their perception of health related quality of life (HRQL). This effect was not measured in this study.
All of the PLCO participants entering into the study that were randomized during the months of April 1997 through September 1997 were included in the sample. Those randomized in April 1997 were mailed a baseline questionnaire prior to their first screening exams (T0) that began in November 1997, continuing until the sample randomized in September 1997 were mailed a baseline questionnaire prior to their first screening exam in April 1998. The first follow-up study was conducted after the baseline study, six months prior to T1 (Fig. 1), with those participants randomized in April 1997. This continued until the last monthly sample of PLCO participants randomized in September 1997 were mailed a first wave follow-up questionnaire. The second follow-up was done for all participants during April through June of 2002, approximately three and one-half years after the first wave follow-up.
MATERIALS AND METHODS
Sample
At baseline, all 899 newly randomized PLCO-Hawaii participants (both in the screened and control group) during the months of April through September 1997 were selected with 674 responding to the mail-out, mail-back questionnaire (a 75% return rate). The baseline study was done prior to participant initial screening in order to determine their ‘prior to screening’ status. The first follow-up was sent out after the initial screening results (T0) were known by the participants and prior to T1. Of the 674 respondents participating in the baseline study, all were mailed the same questionnaire at 1st follow-up and 577 chose to respond (an 86% return rate). The second follow-up was sent out in 2002 and 522 participants chose to respond (284 screenees and 238 controls), which is a 90% return rate. This allowed participants to have received three annual screening exams during the second follow-up period.
With such a high return rate to all three surveys, it is unlikely that there was a selection bias introduced by non-response. This report focuses on the responses provided by the 522 participants who completed all three surveys, showing the changes in responses provided to the survey instrument over time.
The actual PLCO screenings were: PSA and digital rectal exams (yearly) for prostate cancer screening, trans-vaginal ultrasound and CA125 (yearly) for ovarian cancer screening, x-ray (yearly) for lung cancer screening and flexible sigmoidoscopy for colon cancer screening (years 1 and 6). Each PLCO participant randomized for screening received at least one screening exam each year. PLCO participants randomized to the control group were not screened, but received an annual follow-up questionnaire from PLCO. All those selected for this study received the same SF-36 questionnaire (instrument) on the schedule described previously.
Instrument
The instrument employed in this study is commonly known as the SF-36 Health Survey. It was originally developed for the Medical Outcomes Study and has been extensively tested and validated in various studies of chronic disease as well as with older populations. Over half of the Medical Outcomes Study population was over 60 years of age, about 40% were eligible for Medicare and all had one or more chronic conditions (Ware, 1993). This profile is similar to that of PLCO population. The content of the SF-36 questions reflect common themes found in health insurance literature, thus allowing for comparison with findings of other studies. The SF-36 allows researchers to measure eight different interrelated aspects of health. These include: physical functioning, that measures limitations in performing physical activities; role-physical, that measures problems with work or other daily activities as a result of physical health; bodily pain, that measures limitations due to pain; general health, that evaluates the person’s overall health; vitality, that measures the extent to which a person feels full of pep and energy or feels tired and worn out; social functioning measures the extent to which the person can perform normal social activities without interference due to physical or emotional problems; role emotional, that determines the extent to which the person experiences problems with work or other daily activities, due to physical or emotional problems; mental health, that measures the person’s feeling of being happy and calm or nervous and depressed. The final question, health transition, measures whether the person believes their health is much worse or much better now than a year ago (Ware, 1997). Each of the eight SF-36 scales have been standardized and can be compared with the health profile for U.S. adults as well as norms for other specific sub-groups of the population ago (Ware, 1997).
In this study, for purposes of analytical simplicity, the eight SF-36 subscales, described above, have been consolidated into two subscales, a Physical Health Summary scale (PCS) and a Mental Health Summary scale (MCS). The rationale and procedure for accomplishing this consolidation is described in “SF-36 Physical and Mental Health Summary Scales: A User’s Manual” (Ware et al., 1994). These two summary scales are standardized to the U.S. Population with a z-score transformation and norm based scoring, where the mean is 50 and the standard deviation is 10. These two subscales are the independent variables for the health related quality of life measures.
Statistical Analysis
Data was entered into an Access database and then converted into an SPSS data file for analysis. The data was scored into the SF-36 eight scales which were then transformed to z-scores which were then used to consolidate the eight scores into the two summary scales (PCS and MCS). Tabulations were computed showing means and standard deviations by various demographic characteristics and by case and control. In addition, a multi-variate analysis of variance was computed for screenees with the results at baseline and for 1st and 2nd follow-up to show time trends for each of the eight scales. In addition, the data set was screened for cases with abnormal screening outcomes at each of the two-study follow-up times and an analysis was conducted to determine the effect of receiving an abnormal screening outcome upon each of the SF-36 summary scales. The detailed results of this analysis are discussed in the Results section of this paper.
RESULTS
Demographic Profile of Respondents
Table 1 shows the demographic distribution of the sample by gender, age and ethnicity (race) for participants in the screened and control group. The sample distribution by gender shows a slightly higher proportion of males than females, for both groups. The distribution by age group shows the two youngest age groups (55-64 and 65-74) are about equal in size and the oldest age group is much smaller (75 +). This pattern is consistent for both groups. The distribution by ethnicity shows the Caucasians are most numerous, followed by Japanese. The Chinese sample is small, as is the sample of other ethnic groups. The same distribution pattern is found in both groups. It appears from Table 1 that the sample participants in the screened and control groups are quite similar in their distribution by gender, age and ethnicity.
Abnormal (Positive) Screening Results by Time
The following discussion deals only with the participants in the screened group. The screening results were obtained from the PLCO-Hawaii office, for each of the study participants. These included the type of cancer screen conducted at baseline and at each annual visit during the entire study period. As mentioned earlier, male subjects were screened for prostate, lung and colon cancer and females were screened for ovarian, lung and colon cancer. Each case had one or more screening exams at each annual visit. The results of each annual screening were made known to the participant. If there was an abnormal (positive) screen, then verification was made with the participant’s medical record and/or physician. This verification process took a while to complete and so by the time the participant received the SF-36 questionnaire, they only knew they had one or more abnormal screening results, even though they might not have been verified.
| Table 1: | Distribution of sample by study group, gender, age and ethnicity |
 | |
| Source: PLCO-Hawaii SF-36 Ancillary Study | |
At the study baseline, the participants had not yet been screened for the first time and therefore no abnormal screens were possible. At the first study follow-up, one annual screening had been done and so it was possible for participants to have one or more abnormal screens, depending on how many exams they had. Between the first and second follow-ups of the SF-36 questionnaire, an additional three annual screenings had been received by participants and so additional abnormal results were possible. Table 2 shows the distribution of participants by number of total abnormal screens during the entire study and at each time, i.e., 1st SF-36 follow-up and 2nd SF-36 follow-up.
There are a total of 282 participants followed from the study baseline to the first follow-up and through the second follow-up. Of these, 193 had no abnormal screening results during the entire study. Thus 68.4% of the studied participants received no abnormal screening results at all. Some 89 participants (or 31.6%) had one or more abnormal screening results during the study period (i.e., through the 2nd follow-up). During the first follow-up screening, only 40 had one abnormal screen and one had two abnormal screens and the rest of the participants (241) had no abnormal screening results at that time. By the time of the second follow-up, which was three years later, a total of 89 participants had been screened abnormal a total of 162 times. This means that each participant receiving an abnormal screen during that time had an average of 1.82 abnormal screens.
Table 2 shows the distribution of abnormal screens. At the first follow-up only one had been screened twice with the rest only once. By the second follow-up two had an abnormal screen six times during the 1st and 2nd follow-up period. Two participants had abnormal screens five times, seven had abnormal screens four times, ten had abnormal screens three times, 14 had abnormal screens two times and 54 had an abnormal screen only once.
Table 2 shows a progressive increase in the number of participants receiving abnormal screens over time and, for those who have abnormal screens, a progression over time of the number of abnormal screens.
| Table 2: | SF-36 ancillary study, number of abnormal screens at first (T0) and at second follow-up (T0-T4) |
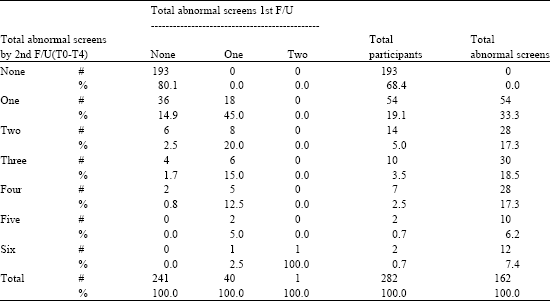 | |
| Note: There were no abnormal screens at baseline (SF-36 Study), * Of the 89 participants with abnormal screens (T0-T4), there were a total of 162 abnormal screens or an average of 1.82 abnormal screens during the 4-year period for those with at least one abnormal screen | |
The number of abnormal screens received at each follow-up will constitute the independent variable for this study. The number of abnormal screens is posited to have a negative effect on the Health Related Quality of Life (i.e., PCS and MCS) and cases with more abnormal screens are expected to have a lower self-perceived Health Related Quality of Life than those with fewer abnormal screens or those cases with no abnormal screens, at each follow-up. As the number of abnormal screens increases from 1st to 2nd follow-up, this is expected to result in the deterioration of self-perceived Health Related Quality of Life, i.e., the reduction in the PCS and MCS scales.
In addition to the information about the abnormal screening results obtained at each annual examination, the PLCO participant records also showed if there were any cancer diagnoses and for which sites. Table 3 shows the distribution of diagnosed cancers by site and PLCO study year. As of April 2002 at the end of the follow-up period for this study the PLCO participants in this study had gone through year T4 of the PLCO study. As of this date, only five PLCO cancers had been diagnosed among the 89 participants that had received an abnormal cancer screening result during that time. There were two prostate cancers, one lung cancer, two ovarian cancers, no colorectal cancers. In addition, there were seven other cancers recorded, that were not diagnosed in the PLCO protocol. As can be seen on Table 3, three cancers were diagnosed at T0, none at T1, two at T2 and none at T3 and T4. It is likely that additional diagnoses will be recorded as verification is received from the patient’s medical records.
Analysis of SF-36 Summary Scales by Demographic Characteristics, Study Group and Time
The two dependent variables consist of two SF-36 summary scales, i.e., PCS for physical health and MCS for mental health, i.e., self-perceived as of the baseline, first follow-up and second follow-up. The independent variables are study group and demographic characteristics, i.e., gender, age and ethnicity.
Table 4, shows the results for the physical summary scale (PCS) for cases, participants who are screened and controls over time by each demographic characteristic. The PCS is normalized and standardized to a mean of 50 and standard deviation of 10.
The results of Table 4a show slight variations above and below the mean, all within one standard deviation. There are no significant trends over time or between participants in the screened and control group or across various categories of gender, age or ethnicity.
| Table 3: | SF-36 study diagnosed cancers by site and PLCO study year |
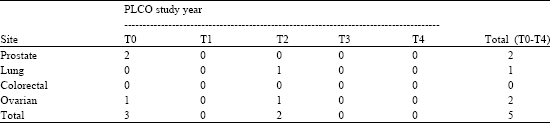 | |
| Note: These cancers diagnosed by April 2002 (study year T4). It is expected that eventually more of the abnormal screens will become diagnosed cancers, due to the lag time from screening to diagnosis. There is no measure of false positives at the time 0. There were 7 additional non-PLCO diagnosed cancers identified during this time. | |
| Source: PLCO-Hawaii | |
| Table 4a: | Physical and mental summary scales by time, study group, gender, age and ethnicity |
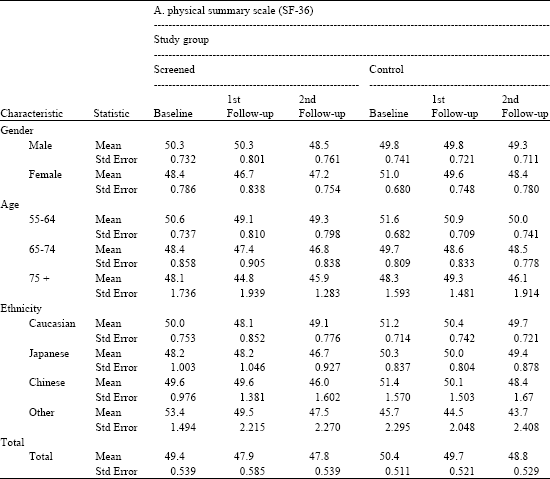 | |
Source: PLCO-Hawaii SF-36 Ancillary Study. Note: No significant differences in means (p≤0 .05) | |
Table 4b shows the results for the Mental Summary Scale (MCS) over time by each demographic characteristic. As with PCS, the MCS is normalized and standardized to a mean of 50 and standard deviation of 10. The results show slight variations above and below the mean, all within one standard deviation. With only one exception, there are no significant trends over time or between participants in the two allocated groups or across various categories of gender, age or ethnicity. There are, however, two means in the oldest age group (age 75+) cases for baseline and 1st follow-up that are significantly lower than the MCS means for the other two age groups. This indicates a slight downward trend in self-perceived mental health for the oldest age screened participants (but not the controls). Even so, the MCS means for the oldest age group (age 75+) over time do not differ significantly nor do they differ when comparing the two study groups.
Generally, we see for both the physical and the mental SF-36 summary scales no significant differences between the study groups, no significant differences over time, no significant differences between gender groups, or ethnic groups and only with one exception, i.e., the mental summary scale for screened group participants in the oldest age group, no significant differences between age groups.
Analysis of Abnormal Screening Results and SF-36 Summary Scales
The two dependent variables consist of two SF-36 summary scales, i.e., PCS for physical health and MCS for mental health, i.e., self-perceived as of the baseline, first follow-up and second follow-up.
| Table 4b: | Physical and mental summary scales by time, study group, gender, age and ethnicity |
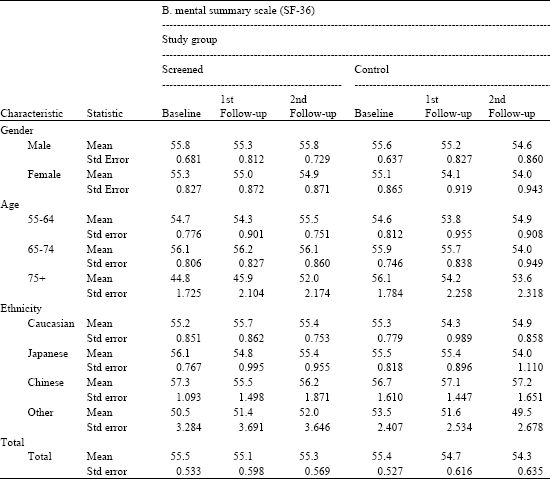 | |
| Source: PLCO-Hawaii SF-36 ancillary study Note: Significantly lower means in bold face (p = <0 .05) | |
The independent variables are the number of abnormal (positive) screening results a case received at each of those times. Since no screening had started for the PLCO participants at the baseline of this study, no abnormal screening results were possible. However, by the first follow-up one annual follow-up screening, with one or more screening exams, made it possible to have one or more abnormal screening results.
Table 5 below shows only one participant had two abnormal screening results and the other 40 had one abnormal screening result, with the remaining 241 participants having no abnormal screening results at the first follow-up. Table 5 shows the corresponding PCS mean scores for participants with one and two abnormal screening results. An ANOVA was conducted which resulted in a non-significant effect of the number of abnormal screening results upon the PCS and MCS score for the first follow-up. There is no significant trend in PCS and MCS means by number of abnormal screening results.
The second follow-up conducted three years after the first follow-up shows as many as six abnormal screening results picked up by that time. Mean PCS and MCS scores were computed for each level of the abnormal screening results, from 0 to 6 and an ANOVA was conducted using the number of abnormal screens as the independent variable and the PCS and MCS scores as the dependent variables. Table 5 indicates that there was no significant effect of the number of abnormal screens upon PCS and MCS.
| Table 5: | SF-36, PCS and MCS number, means and standard errors by number of abnormal screens by study follow-up |
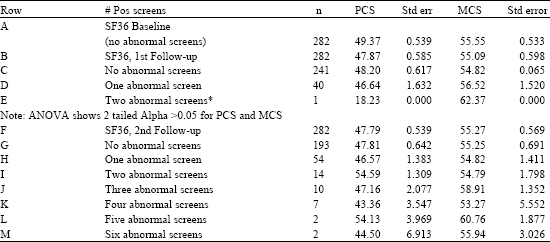 | |
| Note: ANOVA shows 2 tailed Alpha = >0.05 for PCS and MCS. *Where only 1 case is found in the cell, the case specific PCS or MCS score is used | |
| Table 6: | SF-36 PCS and MCS means by whether any abnormal screen and/or diagnosed cancer |
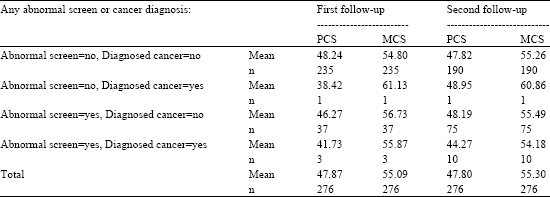 | |
| Note: Comparisons within each follow-up between means for MCS or PCS not significant at p = <0.05. | |
| Source: PLCO-Hawaii | |
This indicates that the number of abnormal screenings (at both the first and second follow-up) is neither a significant determinant of the case’s self-perceived physical health status nor a significant determinant of self-perceived mental health status.
An additional consideration was given to effects of having a cancer diagnosis. Table 6 shows the PCS and MCS mean scores for participants at each follow-up. The categories are arranged with those that are expected to have the least negative effect on the scores (the top two) to the category at the bottom with the greatest negative effect. The third category from the top is expected to have an intermediate effect compared with others, i.e., two at the top and at the bottom. The least expected negative effect is: no abnormal screen and no cancer diagnosis (235 cases at 1st follow-up and 190 cases at 2nd follow-up) and no abnormal screen and has a diagnosis of cancer (just 1 case at first and 1 at 2nd follow-up). Next are those with one or more abnormal screens but no diagnosed cancer (37 cases). The last category (at the bottom) is expected to have the greatest negative effect, includes those with both one or more abnormal screens and one or more diagnosed cancers (3 cases at 1st follow-up and 10 cases at 2nd follow-up). In observing the table, no such overall trend is observed. The fact that there is no overall trend is no doubt the result of too few subjects in some of the categories to show significance. It appears, from Table 6, that the data are insufficient to show any added effect of having a cancer diagnosis over the effect of having a positive screening result upon the self-perceived HRQL.
DISCUSSION
The data show no significant trends in the SF-36 physical or mental summary scales when comparing screened participants with controls, when comparing across time, i.e., baseline, 1st follow-up and 2nd follow-up and when comparing across the categories of the demographic characteristics (with the exception of the oldest age screened participants having a low PCS). All physical and mental summary scale means are within one standard deviation, i.e., 10, of the standardized mean, i.e., 50.
The data show an increasing number of abnormal screening results over time. This indicates increasing pressure for a downward trend in self-perceived HRQL, i.e., the SF-36 summary scales. It is observed, however, that there is neither a downward trend in PCS (physical health) nor in MCS (mental health) mean scores. This is observed at each follow-up.
The number of abnormal cancer screening results increased over time, due to the number of screening exams administered to PLCO participants. Independent of the number of abnormal screenings, there seems to be no significant trend in Health Related Quality of Life over time. While the effect of aging of the participants over time could be expected to produce a decline in Health Related Quality of Life, since the study was conducted over a relatively short time span the effect of aging, while showing slight declines in mean scores, when taking the increasing number of abnormal screening results into account, these changes are not statistically significant.
As persons experience increasing evidence of cancer over time, manifest by the accumulation of abnormal screening outcomes, it would be expected that the self-perceived Health Related Quality of Life would be negatively affected. This does not seem to be the case with self-perceived physical health (PCS) or for self-perceived mental health (MCS). Thus, the main hypothesis of this study was not supported, i.e., self-perceived physical health and mental health are negatively affected by knowledge of an abnormal cancer-screening outcome. This means that there are no negative effects in a person’s Health Related Quality of Life resulting from participation in cancer screening.
In considering whether having one or more cancer diagnoses has an independent effect on self-perceived Health Related Quality of Life, the number of cancer diagnoses verified during the study was insufficient (only 5 PLCO diagnosed cancers plus 7 others) to show a significant trend in effect upon PCS or MCS mean scores.
It may be that eventually, as the disease progresses, when the patient receives treatment for the disease, this would have an effect on their self-perceived physical health status and self-perceived mental health status. However, this might be the subject of additional research, as it was not possible within the scope of this study to address this issue.
ACKNOWLEDGMENTS
The author wishes to thank all of the PLCO Participants who contributed to this study, especially those who responded to the survey all three times! The PLCO Staff, especially Louisa Turner and Vicki Jenkins plus the PHRI staff, especially Ruth Baldino, Vicki Shambaugh, Wally Izumigawa and Geri Lee were instrumental in initiating the project and seeing it through and to Sue Kohler for her administrative support. Special thanks goes to the late Dr. Fred Gilbert, M.D. who initially conceived the project and Dr. Kim Kimminau who wrote the first funding proposal through PHRI to NCI. Dr. Lance Yokochi, M.D., who is the PLCO-Hawaii principal investigator has supported the project since its inception. Dr. John Gohagan and Dr. Philip Prorok of NCI have supported the funding of the project and encouraged the extension of the project to allow for an additional follow-up. Dr. Anthony Miller was very helpful in reviewing the initial protocol and in reviewing the study in progress. Without the combined efforts of these dedicated persons, this research project would not have been possible. The author also expresses appreciation to Barbara Ankersmit, President of QMark Research and Polling for supporting this project.
REFERENCES
- Vernon, S.W., B.C. Tilley, A.V. Neale and L. Steinfeldt, 1985. Ethnicity survival and delay in seeking treatment for symptoms of breast cancer. Cancer, 55: 1563-1571.
PubMed








