Research Article
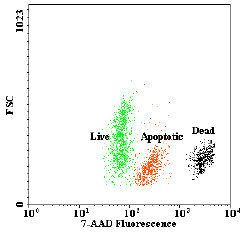
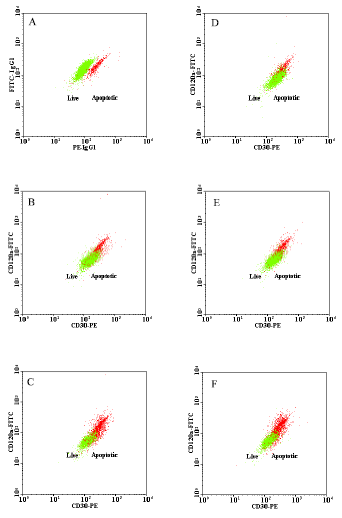
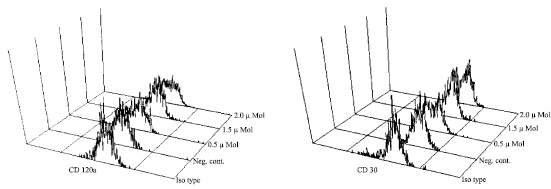
Evaluation of Apoptosis Induced by Arsenic Trioxide Through TNFR-I and CD30 Pathways in Acute Promyelocytic Leukemia Patient with t (15;17) Translocation
Hematology, Oncology and BMT Research Center, Tehran University of Medical Science, Tehran, Iran
K. Alimoghadam
Hematology, Oncology and BMT Research Center, Tehran University of Medical Science, Tehran, Iran
S. Kaviani
Department of Hematology, Faculty of Medicine, Tarbiat Modares University, Tehran, Iran
A. Ghavamzadeh
Hematology, Oncology and BMT Research Center, Tehran University of Medical Science, Tehran, Iran
M. Djahani
Hematology, Oncology and BMT Research Center, Tehran University of Medical Science, Tehran, Iran
L. Moezzi
Hematology, Oncology and BMT Research Center, Tehran University of Medical Science, Tehran, Iran