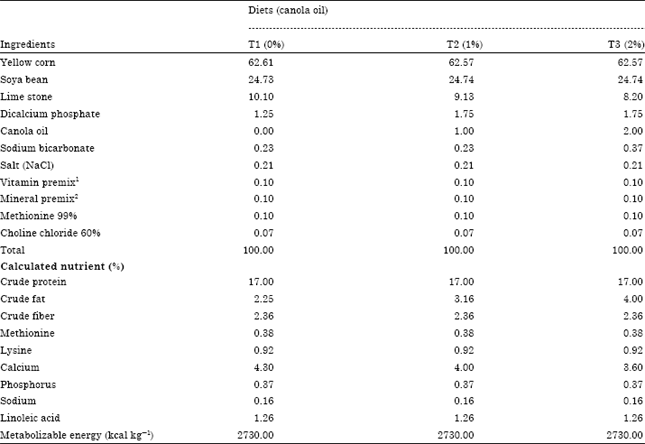
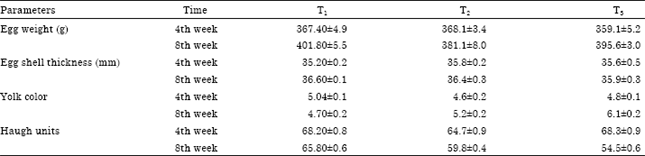
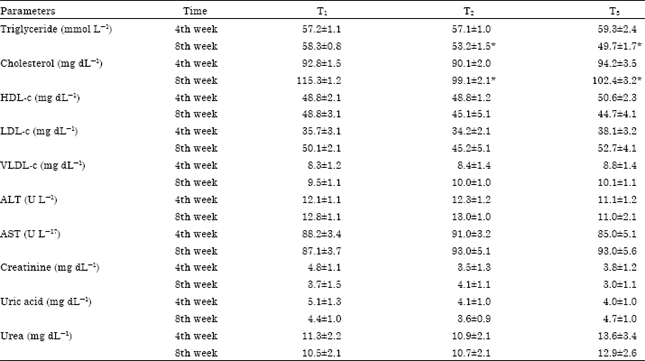
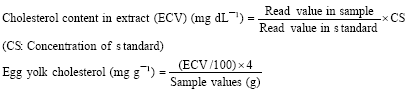Research Article
Effect of Dietary Supplementation of Canola Oil on Egg Production, Quality and Biochemistry of Egg Yolk and Plasma of Laying Hen
Department of Physiology, Biochemistry and Pharmacology (Physiology), College of Veterinary Medicine and Animal Resources, King Faisal University, Al-Ahsa, Saudi Arabia
K.A. Al-Busadah
Department of Physiology, Biochemistry and Pharmacology (Physiology), College of Veterinary Medicine and Animal Resources, King Faisal University, Al-Ahsa, Saudi Arabia
S.M. El-Bahr
Department of Biochemistry, Faculty of Veterinary Medicine, Alexandria University, Egypt