Research Article
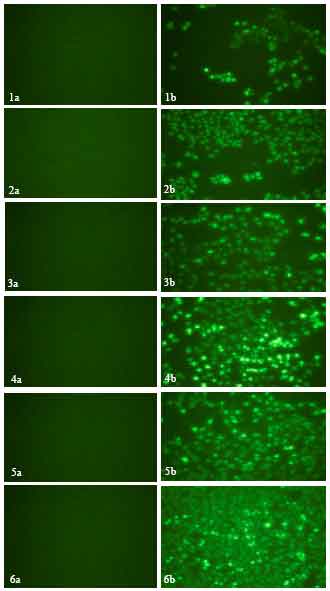
Apoptosis Study on the Effect of PMF on Different Cancer Cells
Department of Biology,
S.A. Rahimaldeen
Department of Biochemistry, Faculty of Science, King Abdul Aziz University, Jeddah, Saudi Arabia
J.S. AL-Amri
Department of Biology,