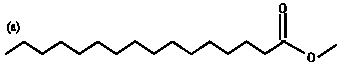
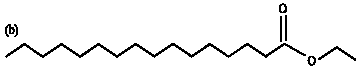
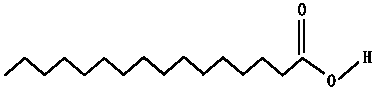
Research Article
Chemical Constituents and Hemolytic Activity of Macrotyloma uniflorum L.
Department of Chemistry, Faculty of Science, University of Chittagong, Chittagong-4331, Bangladesh
G. Mostafa
Department of Chemistry, Faculty of Science, Chandpur Government University College, Chandpur, Bangladesh
E. Huq
Department of Chemistry, Faculty of Engineering, Bangladesh University of Engineering and Technology, Dhaka-1000, Bangladesh
N. Nahar
Department of Chemistry, Faculty of Science, University of Dhaka, Dhaka-1000, Bangladesh
Y. Ozeki
Department of Biochemistry, International Graduate School of Arts and Sciences, Yokohama City University, 22-2 Seto, Kanazawa-Ku, Yokohama 236-0027, Japan