Research Article
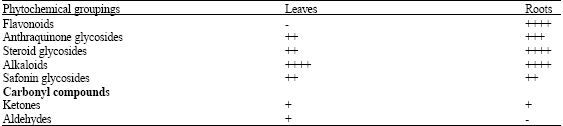
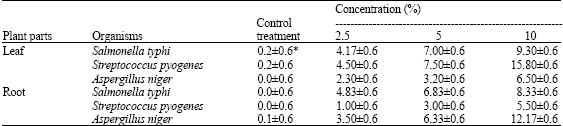
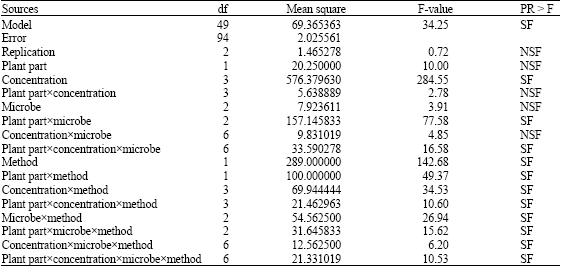
Effects of Leaf and Root Extracts of Aspilia Plant (Aspilia mossambicensis) (Oliv) Wild.on Some Selected Micro-Organisms
Department of Botany and Horticulture,
J.A. Ogur
Department of Chemistry, Faculty of Science, Maseno University, P.O. Box 333, Maseno, Kenya
P.M. Muema
Department of Chemistry, Faculty of Science, Maseno University, P.O. Box 333, Maseno, Kenya