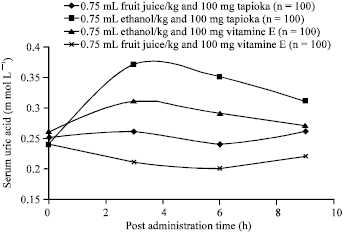
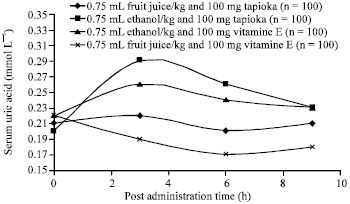
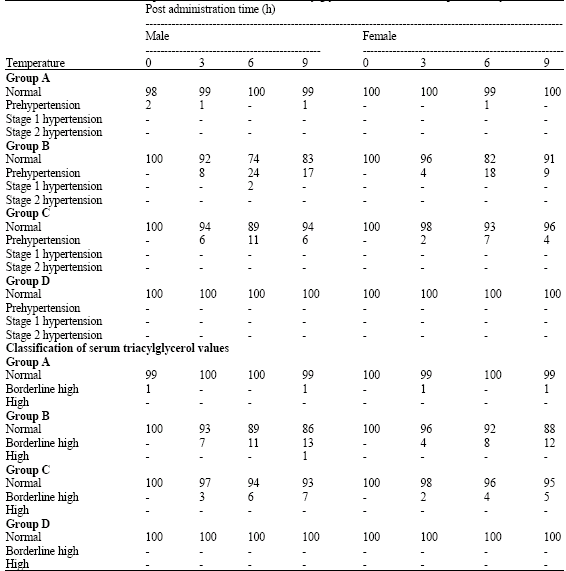
Research Article
Effect of Vitamin E on Ethanol-induced Increase in Some Cardiovascular Parameters and Blood Uric Acid Levels in Man
Department of Biochemistry,
U.E. Uzuegbu
Department of Biochemistry,
E.K. Nwangwa
Department of Physiology,
G.E. Umukoro
Department of Medical Microbiology, Delta State University, Abraka, Nigeria
I.R. Aninyei
Department of Physiology,