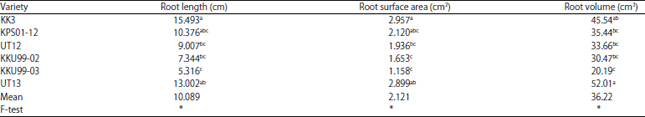
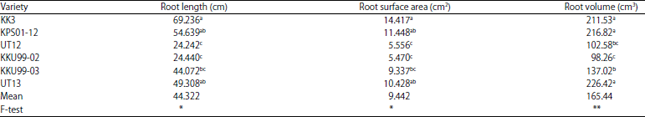
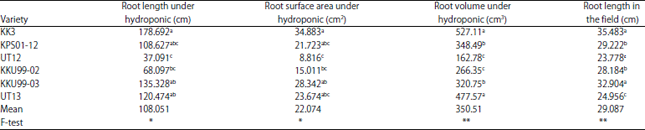
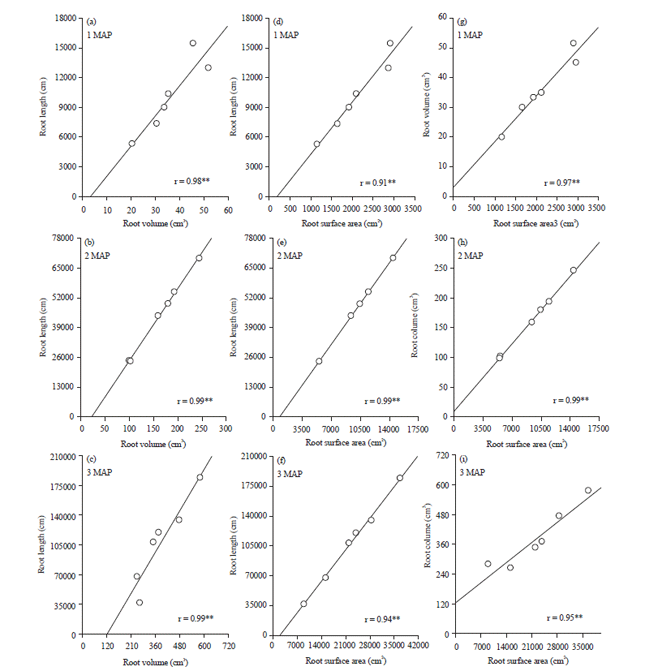
Research Article
Determination of Early Effective Screening Date on Phenotyping Sugarcane Roots under Hydroponics Condition
Department of Agronomy, Faculty of Agriculture, Khon Kaen University, Muang, 40002 Khon Kaen, Thailand
Jidapa Khonghintaisong
Department of Agronomy, Faculty of Agriculture, Khon Kaen University, Muang, 40002 Khon Kaen, Thailand
Santimaitree Gonkhamdee
Department of Agronomy, Faculty of Agriculture, Khon Kaen University, Muang, 40002 Khon Kaen, Thailand
Patcharin Songsri
Department of Agronomy, Faculty of Agriculture, Khon Kaen University, Muang, 40002 Khon Kaen, Thailand
Nuntawoot Jongrungklang
Department of Agronomy, Faculty of Agriculture, Khon Kaen University, Muang, 40002 Khon Kaen, Thailand
LiveDNA: 66.25059