ABSTRACT
Background and Objective: Cichorium intybus Linn. (C. intybus L.) has been known in traditional medicine since ancient times, therefore, the present study aimed to investigate the phytochemical composition, antioxidant activity and antimicrobial potentiality of aqueous and methanol extracts of aerial parts of C. intybus L. Materials and Methods: Total phenolic, flavonoids, alkaloids, tannins and saponins were assessed as the Mean±SD of three readings in the extracts of the aerial parts of C. intybus L. The extracts were examined for their antioxidant and antimicrobial activities. Results: The methanol extract was found to possess a comparatively higher values of total phenolic, flavonoids, alkaloids and tannins than aqueous extract except saponins which seem higher in aqueous than methanol extract. The methanol extract exhibited a relatively high antioxidant activity as it showed comparatively low values of IC50 for 2,2-Diphenyl-1-picrylhydrazyl (DPPH) inhibition than ascorbic acid (standard) and the aqueous extract. Both extracts were screened for their antimicrobial activity against 11 pathogenic microbial strains and showed significant inhibition zones. The methanol extract showed broader antimicrobial spectrum than the aqueous extract. Conclusion: The results of this study indicated that the aerial parts extracts of C. intybus L. have good phytochemical composition, good antioxidant activity and antimicrobial potentiality.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijb.2018.24.29
URL: https://scialert.net/abstract/?doi=ijb.2018.24.29
INTRODUCTION
Genus Cichorium comprises 6 species that are widely distributed in Africa, Asia, Europe, Australia and America1-4. Cichorium intybus Linn, family Asteraceae (compositae), is commonly known as chicory. It is an erect herbaceous annual herb, around 4-110 cm in height with a strong fleshy taproot, solitary erect stem and large basal leaves roseate, ovate to lanceolate, attenuate into a petiole-like basal portion1,4. The plant was used traditionally for the treatment of diarrhea, fever, jaundice, gallstones, prostate, pulmonary disease, cough, cancer, liver complaints and mild digestive disorders4,5.
Cichorium intybus is characterized by presence of variety of compounds like sesquiterpene lactones, derivatives of caffeic acid, inulin, proteins, hydroxycoumarins, phenolic, flavonoids, alkaloids, terpenes, essential and volatile oils, coumarins and vitamins6-10, that possess many pharmacological effects including hepatoprotective, gastroprotective, cardiac, anti-oxidant, anti-cancer, anti-diabetic, anti-microbial, anti-protozoal and anti-inflammatory activity4,6-13.
The non-nutritive plant phytochemicals, like phenolic, flavonoids, alkaloids, tannins and vitamins play a crucial role in many functions in human body as these compounds possess antioxidant properties and protect macromolecules like lipids, proteins and nucleic acids against oxidative damage. The reaction of the free radicals with these macromolecules cause apoptosis, that leads to many neurological, cardiovascular and some physiological disorders3,6,14.
Some studies deals with C. intybus as Nandagopal and Kumari7 and Basa’ar et al.15 screen qualitatively some phytochemical compounds in C. intybus root and determine its antimicrobial activities. Regarding C. intybus seeds, Mehmood et al.16 investigate the antioxidant and antimicrobial activities as well as the qualitative survey for some secondary metabolites. Shad et al.6 study the antioxidant activities and some metabolites in the plant root, stem leaves and seed whereas, Abbas et al.3 and Kaur et al.17 studied the antioxidant and some phytochemical constitutes in C. intybus leaves.
Therefore, the present study was designed to demonstrate the phytochemical constituents, antimicrobial and antioxidant activities of the aqueous and methanol extracts of the dried aerial parts of C. intybus.
MATERIALS AND METHODS
Plant materials: The aerial parts of Cichorium intybus obtained from fields of Misan governorate, Iraq were air dried in shade for 5 days (till complete dryness) during September, 2017.
Instruments and chemicals: Most of the chemicals used were laboratory-grade compounds purchased from Merck. DPPH was obtained from Sigma Chemical. All other reagents used were of analytical grade.
Preparation of the extracts: Five grams of the air dried plant material were extracted using 80% methanol upon shaking for 2 h at 200 rpm and using distilled water at 70°C upon shaking for 30 min. The extracts were filtered using Whatman No.1 filter paper and then dried using rotary evaporator followed by lyophilizer for complete drying. The crude extracts obtained were dissolved in Dimethyl Sulfoxide (DMSO) and kept in an air-tight containers stored in a refrigerator for use.
Phytochemical constituents
Determination of total phenolic compounds: The determination of total phenolic compounds in the extracts was carried out using Folin Ciocalteu assay according to the method described by Wolfe et al.18. The total phenolic compounds were reported as gram of gallic acid equivalents/100 gram of the dried plant material (g/100 g).
Determination of total flavonoids content: The total flavonoids were determined using aluminum chloride colorimetric assay developed by Zhishen et al.19. The total flavonoids content were expressed as g/100 g dried plant (gram catechins equivalent per 100 gram of the dried plant material).
Total alkaloids: The total alkaloid contents were measured using 1,10-Phenanthroline method described by Singh et al.20. The total alkaloids content was expressed as g/100 g dried plant (gram colchicines equivalent/100 gram dried plant material).
Total tannins: Tannin content was determined by vanillin hydrochloride method of Sadasivam and Maickam21. The total tannins were reported as g/100 g dried plant (gram of tannic acid equivalent/100 gram of the dried plant material).
Total saponins: Saponins content was determined using the method of Obadoni and Ochuko22. The saponins content were calculated as g/100 g dried plant (gram saponins/100 gram dried plant material).
Antioxidant activity
Determination of the radical scavenging activity using DPPH (2,2-Diphenyl picrylhydrazyl) assay: The antioxidant activities of the methanol and aqueous extracts from the air dried aerial parts of Cichorium intybus were assessed using the radical scavenging assay of stable DPPH. The scavenging activity of the plant extracts on DPPH radical was estimated according to the method described by Kitts et al.23. One milliliter of suitable concentration of the tested extracts was added to 1 mL of DPPH (0.135 mM) and kept in dark for 30 min. The absorbance was measured at 517 nm and the percent of the remaining DPPH of each of the tested concentrations was estimated and plotted against the concentrations of the used extracts. The concentration of antioxidant that is needed to decrease the initial DPPH concentration by 50% (IC50) was calculated for each extract. Ascorbic acid was used as a standard agent.
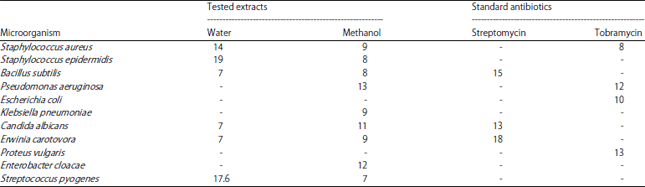
Antimicrobial activity: Antimicrobial activity was tested by agar disc diffusion method. The agar plates were inoculated with inoculum containing 106 microbial cells of the different selected bacterial strains using a streak plate method. Six millimeter filter paper discs were soaked overnight in the prepared plant extracts and loaded on the agar. After incubation at 37°C for 24 h, the antimicrobial efficiencies of the extracts were assessed on the basis of the inhibition zone24. Antimicrobial activity was tested against ten pathogenic bacterial strains namely, Escherichia coli, Staphylococcus aureus, Staphylococcus epidermidis, Bacillus subtilis, Pseudomonas aeruginosa, Klebsiella pneumoniae, Erwinia carotovora, Proteus vulgaris, Enterobacter cloacae and Streptococcus pyogenes, in addition to one fungal pathogenic strain: Candida albicans. The bacterial strains were obtained from the Laboratory of genetic engineering and biotechnology unit at Faculty of Science, Mansoura University in Egypt. The results were compared with tobramycin and streptomycin.
Statistical analysis: The phytochemical constituents (total phenolic, total flavonoids, total alkaloids, tannins and saponins) were expressed as the Mean±SD of three readings.
RESULTS
Phytochemical analysis: The data obtained in Table 1 revealed that Cichorium intybus extracts of the aerial parts possess a variety of different phytochemical compounds. The investigated compounds (total phenolic, total flavonoids, total alkaloids and total tannins) were higher in methanol extract of the plant aerial parts than those found in the water extract of the plant aerial parts.
The total phenolic content in C. intybus dried aerial parts was ranged from 1.01 and 2.04 g/100 g dried plant in water and methanol extracts, respectively. The total flavonoids content was 0.26 and 0.54 g/100 g dried plant in the water and methanol extracts, respectively. Regarding the total alkaloids content, it was 0.50 and 0.99 g/100 g dried plant in water and methanol extracts, respectively. Comparing to the total phenolic, total flavonoids and total alkaloids, the aerial parts of C. intybus found to possess a relatively high content of tannins which attained 1.61 and 3.02 g/100 g dried plant in water and methanol extracts, respectively.
Saponins contents showed opposite trend to the other estimated metabolites, where it was higher in water extracts of the plant aerial parts than that found in the methanol extract. It was ranged 1.14 and 0.65 g/100 g dried plant in the water and extracts, respectively.
Evaluation of the antioxidant activity
The free radical scavenging activity using DPPH assay: The concentration of an antioxidant needed to decrease the initial DPPH concentration by 50% (IC50) is a parameter widely adopted for measuring the antioxidant activity, where the lower the IC50, the higher the antioxidant power. Ascorbic acid was employed as standard compound in this assay. The illustrated data in Fig. 1 showed that the antioxidant activity of the C. intybus was 0.01 mg mL–1 in methanol extract, 0.11 mg mL–1 in water extract and 0.02 mg mL–1 for the ascorbic acid.
| Table 1: | Phytochemical content of aqueous and methanol extracts of Cichorium intybus aerial parts. The mean values indicated±standard deviation |
 | |
| Table 2: | Antimicrobial activity of aqueous and methanol extracts of Cichorium intybus aerial parts using disc diffusion assay |
 | |
Values indicate zone of inhibition in mm and include filter paper disk diameter (6 mm), -: No inhibition | |
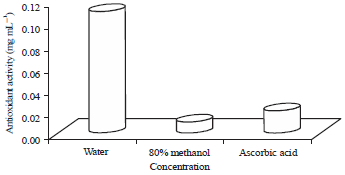 | |
| Fig. 1: | Antioxidant activity of aqueous and methanol extracts of Cichorium intybus aerial parts using DPPH (2,2-Diphenyl picrylhydrazyl) assay |
Evaluation of the antimicrobial potential: It is obvious from the obtained results in Table 2 that the antimicrobial activity of C. intybus aerial parts of aqueous and methanol extracts tested against eleven pathogenic microorganisms showed varied trend. Thus, the methanol extract exhibited broader antimicrobial spectrum than the aqueous extract comparing with the standard antibiotics gentamicin and tobramycin. The water extract showed stronger activity against Staphylococcus epidermidis (19 mm), Streptococcus pyogenes (17.6 mm) and Staphylococcus aureus (14 mm) even more than that of the standard antibiotics.
DISCUSSION
In this study, quantitative analysis for phytochemical constituents indicated that water and methanol extracts of C. intybus aerial parts poses various secondary metabolites as phenolic, flavonoids, alkaloids, tannins and saponins. Thus, the highest constituents in the studied extracts were tannins followed by phenolic compounds. These results were compatible with those obtained by Shad et al.6, where the phytochemical analysis of stem, leaves and seed showed the same trend of study results but the highest constitutes in the plant root were tannins followed by saponins.
Tannins are polymeric water soluble phenols and are diffuse in herbaceous and woody plants. They have potent antimicrobial activity in addition to their anti-carcinogenic and anti-mutagenic activity25. Phenolic compounds play a crucial role as antioxidants in preventing oxidative cell damage. There is a positive correlation between the total phenolic and the antioxidant activity attributed to the hydroxyl groups present in these compounds due to their hydrogen donating ability6,26-29. Phenolic also possess anti-viral, anti-fungal, anti-inflammatory and anti-mutagenic activities6,30-32. According to Sareedenchai and Zidorn33 phenolic composition varies greatly among the various species of the Cichorieae tribe. For C. intybus, the main reported phenolic compounds were hydroxyl cinnamic acid derivatives, tartaric acid derivatives, flavonols and flavone glycosides, sesquiterpenes lactones, anthocyanins and dicaffeoyl34-40.
Saponins content of the C. intybus aerial parts were higher in the water extract than those found in the 80% methanol extract, in addition saponins were in the highest order after tannins and phenolic content. Shad et al.6 have the same trend but in their study using 20% methanol extracts, the recorded values of saponins in leaves, stem, seed and root were lower than those obtained in this study. This may be due to the increase of the methanol concentration in this study. On the other hand, Abbas et al.3 showed opposite trend, where the flavonoids contents were higher than saponins content in the alcoholic extracts C. intybus leaves. Saponins are the glycosidic compounds found in most of the plants, possess a bitter taste and foaming properties, in addition they have been found to possess antifungal activity and anticarcinogenic3,41.
The estimated alkaloids and flavonoids content in the aerial parts of C. intybus were compatible with those obtained by Shad et al.6. Alkaloids and flavonoids are from polyphenols which acquired attention due to its wide range of biological activities such as antimicrobial, anti-inflammatory, antioxidant, cytotoxicity and anticancer features4-6. Antioxidant activities of phenolic compounds attributed to the presence of hydroxyl groups in their structure29.
The obtained results revealed that methanol extract of the aerial parts showed antioxidant activity higher than that of water and ascorbic acid (standard antioxidant). Shad et al.6 and Abbas et al.3 showed similar trend in the C. intybus leaves which found to contain good free radical scavenging capacity due to their higher DPPH radical inhibition and lower IC50 value. It has been also reported that from the major groups responsible for the antimicrobial activity of the plant extracts are phenolic, flavonoids, alkaloids and lectins38, which recorded in the plant aerial parts.
Antimicrobial activity of C. intybus aerial parts showed that the methanol extract exhibited broader antimicrobial spectrum than the aqueous extract comparing with the standard antibiotics gentamicin and tobramycin. Khakzadihe et al.42 found that there is antibacterial activity C. intybus L. leaves and roots against S. aureus, B. subtilis, E. coli and P. aeruginosa that was in agreement with present study results except for E. coli. In addition, Khakzadihe et al.42 studied antimicrobial effect of chicory leaf extract with different solvents on S. aureus and E. coli and the results indicated that, different solvents of C. intybus leaves against S. aureus and E. coli had no significant difference between. Meanwhile, in the present study, the extracts of the plant aerial parts showed antibacterial activity against S. aureus and were in agreement with the absent of activity against E. coli.
CONCLUSION
The methanol extract of Cichorium intybus aerial parts seems to be richer with the detected phytochemicals than water extract. The presence of the considerable amounts of phytochemicals which are furious source of antioxidants justify the traditional medicinal uses of this plant. Thus, a scientific foundation for the use of this plant in medicine can be established to improve the healthcare of local users.
SIGNIFICANCE STATEMENT
This study confirmed that C. intybus L. aerial parts are rich with many phytochemicals that showed antioxidant and antimicrobial potential. The present study contributes to the effective pharmaceutical formulations and it could be used in improving the human health through its potential role in the antioxidant defense system against free radical generation.
REFERENCES
- Bais, H.P. and G.A. Ravishankar, 2001. Cichorium intybus L-cultivation, processing, utility, value addition and biotechnology, with an emphasis on current status and future prospects. J. Sci. Food Agric., 81: 467-484.
CrossRefDirect Link - Street, R.A., J. Sidana and G. Prinsloo, 2013. Cichorium intybus: Traditional uses, phytochemistry, pharmacology and toxicology. Evidence-Based Complement. Altern. Med., Vol. 2013.
CrossRefDirect Link - Abbas, Z.K., S. Saggu, M.I. Sakeran, N. Zidan, H. Rehman and A.A. Ansari, 2015. Phytochemical, antioxidant and mineral composition of hydroalcoholic extract of chicory (Cichorium intybus L.) leaves. Saudi J. Biol. Sci., 22: 322-326.
CrossRefDirect Link - Al-Snafi, A.E., 2016. Medical importance of Cichorium intybus-a review. IOSR J. Pharm., 6: 41-56.
Direct Link - Shad, M.A., H. Nawaz, T. Rehman and N. Ikram, 2013. Determination of some biochemicals, phytochemicals and antioxidant properties of different parts of Cichorium intybus L.: A comparative study. J. Anim. Plant Sci., 23: 1060-1066.
Direct Link - Zareen, S., M.I. Choudhary, M.N. Akhtar and S.N. Khan, 2008. α-Glucosidase inhibitory activity of triterpenoids from Cichorium intybus. J. Nat. Prod., 71: 910-913.
CrossRefDirect Link - Ahmed, B., S. Khan, M.H. Masoodi and A.H. Siddique, 2008. Anti-hepatotoxic activity of cichotyboside, a sesquiterpene glycoside from the seeds of Cichorium intybus. J. Asian Nat. Prod. Res., 10: 218-223.
CrossRefDirect Link - Shaikh, T., A. Mujum, K. Wasimuzzama and R.A. Rub, 2010. An overview on phytochemical and pharmacological profile of Cichorium intybus Linn. Br. J. Pharmacol., 2: 298-307.
Direct Link - Zaman, R. and S.N. Basar, 2013. A review article of beekhe kasni (Cichorium intybus) its traditional uses and pharmacological actions. Res. J. Pharm. Sci., 2: 1-4.
Direct Link - Mushtaq, A., M. Ahmad and Q. Jabeen, 2013. Pharmacological role of Cichorium intybus as a hepatoprotective agent on the elevated serum marker enzymes level in albino rats intoxicated with nimesulide. Int. J. Cur. Pharmaceut. Res., 5: 25-30.
Direct Link - Mathur, N. and M. Mathur, 2016. Phyto-pharmacology of Cichorium intybus as hepatoprotective agent. Int. J. Pharm. Sci. Rev. Res., 39: 116-124.
Direct Link - Uttara, B., A.V. Singh, P. Zamboni and R.T. Mahajan, 2009. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol., 7: 65-74.
CrossRefPubMedDirect Link - Basa’ar, O., S. Fatema, M. Mohsin and M. Farooqui, 2017. Evaluation of phytochemical and pharmacological properties of Cichorium intybus (L.) based on supercritical fluid extract. Res. J. Pharm. Biol. Chem. Sci., 8: 1857-1866.
Direct Link - Mehmood, N., M. Zubair, K. Rizwan, N. Rasool, M. Shahid and V.U. Ahmad, 2012. Antioxidant, antimicrobial and phytochemical analysis of Cichorium intybus seeds extract and various organic fractions. Iran. J. Pharm. Res., 11: 1145-1151.
Direct Link - Kaur, H.P., I. Singh and N. Singh, 2016. Phytochemical, antioxidant and antibacterial potential of extracts of Cichorium intybus (Chicory). Eur. J. Pharm. Med. Res., 3: 320-332.
Direct Link - Wolfe, K., X. Wu and R.H. Liu, 2003. Antioxidant activity of apple peels. J. Agric. Food Chem., 51: 609-614.
CrossRefDirect Link - Singh, D.K., B. Srivastava and A. Sahu, 2004. Spectrophotometric determination of Rauwolfia alkaloids: Estimation of reserpine in pharmaceuticals. Anal. Sci., 20: 571-573.
CrossRefPubMedDirect Link - Obadoni, B.O. and P.O. Ochuko, 2002. Phytochemical studies and comparative efficacy of the crude extracts of some haemostatic plants in Edo and Delta States of Nigeria. Global J. Pure Appl. Sci., 8: 203-208.
CrossRefDirect Link - Kitts, D.D., Y.V. Yuan, A.N. Wijewickreme and C. Hu, 2000. Antioxidant properties of a North American ginseng extract. Mol. Cell. Biochem., 203: 1-10.
CrossRefPubMedDirect Link - Akiyama, H., K. Fujii, O. Yamasaki, T. Oono and K. Iwatsuki, 2001. Antibacterial action of several tannins against Staphylococcus aureus. J. Antimicrob. Chemother., 48: 487-491.
CrossRefDirect Link - Wang, M., J.E. Simon, I.F. Aviles, K. He, Q.Y. Zheng and Y. Tadmor, 2003. Analysis of antioxidative phenolic compounds in artichoke (Cynara scolymus L.). J. Agric. Food Chem., 51: 601-608.
CrossRefDirect Link - Innocenti, M., S. Gallori, C. Giaccherini, F. Ieri, F.F. Vincieri and N. Mulinacci, 2005. Evaluation of the phenolic content in the aerial parts of different varieties of Cichorium intybus L. J. Agric. Food Chem., 53: 6497-6502.
CrossRefDirect Link - Lobo, V., A. Patil, A. Phatak and N. Chandra, 2010. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev., 4: 118-126.
CrossRefPubMedDirect Link - Fathalla, N., M. Bishr, A.N. Singab and O. Salama, 2015. Phytochemical and biological evaluation of Cichorium intybus L. seeds. IOSR J. Pharm. Biol. Sci., 10: 70-76.
Direct Link - Mares, D., C. Romagnoli, B. Tosi, E. Andreotti, G. Chillemi and F. Poli, 2005. Chicory extracts from Cichorium intybus L. as potential antifungals. Mycopathologia, 160: 85-91.
CrossRefPubMedDirect Link - Dimitrios, B., 2006. Sources of natural phenolic antioxidants. Trends Food Sci. Technol., 17: 505-512.
CrossRefDirect Link - Sareedenchai, V. and C. Zidorn, 2010. Flavonoids as chemosystematic markers in the tribe Cichorieae of the Asteraceae. Biochem. Syst. Ecol., 38: 935-957.
CrossRefDirect Link - Carazzone, C., D. Mascherpa, G. Gazzani and A. Papetti, 2013. Identification of phenolic constituents in red chicory salads (Cichorium intybus) by high-performance liquid chromatography with diode array detection and electrospray ionisation tandem mass spectrometry. Food Chem., 138: 1062-1071.
CrossRefDirect Link - Heimler, D., L. Isolani, P. Vignolini and A. Romani, 2009. Polyphenol content and antiradical activity of Cichorium intybus L. from biodynamic and conventional farming. Food Chem., 114: 765-770.
CrossRefDirect Link - Graziani, G., R. Ferracane, P. Sambo, S. Santagata, C. Nicoletto and V. Fogliano, 2015. Profiling chicory sesquiterpene lactones by high resolution mass spectrometry. Food Res. Int., 67: 193-198.
CrossRefDirect Link - Dupont, M.S., Z. Mondi, G. Willamson and K. Price, 2000. Effect of variety, processing, and storage on the flavonoid glycoside content and composition of lettuce and endive. J. Agric. Food Chem., 48: 3957-3964.
CrossRefDirect Link - Rossetto, M., A. Lante, P. Vanzani, P. Spettoli, M. Scarpa and A. Rigo, 2005. Red chicories as potent scavengers of highly reactive radicals: A study on their phenolic composition and peroxyl radical trapping capacity and efficiency. J. Agric. Food Chem., 53: 8169-8175.
CrossRefDirect Link - Dalar, A. and I. Konczak, 2014. Cichorium intybus from Eastern Anatolia: Phenolic composition, antioxidant and enzyme inhibitory activities. Ind. Crops Prod., 60: 79-85.
CrossRefDirect Link - Sinkovic, L., L. Demsar, D. Znidarcic, R. Vidrih, J. Hribar and D. Treutter, 2015. Phenolic profiles in leaves of chicory cultivars (Cichorium intybus L.) as influenced by organic and mineral fertilizers. Food Chem., 166: 507-513.
CrossRefDirect Link - Rao, A.V. and M.K. Sung, 1995. Saponins as anticarcinogens. J. Nutr., 125: 717S-724S.
PubMedDirect Link - Khakzadihe, M., H. Eslami, M. Rasoulifard, H.M. Zadeh and M.R. Behboudi, 2014. Investigate the antimicrobial effect of chicory leaf extract with different solvents on Staphylococcus aureus and Escherichia coli. Int. J. Biosci., 5: 399-405.
Direct Link








