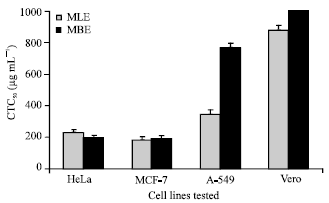
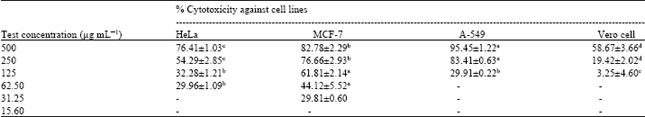
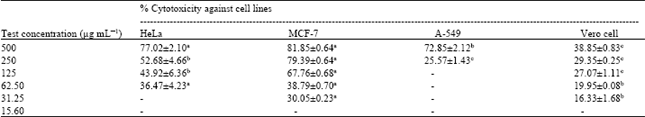
Research Article
In vitro Cytotoxic Evaluation of Hugonia mystax Linn. Leaf and Stem Bark Extracts
Department of Biotechnology, VMKV Engineering College, Periya Seeragapadi, Salem 636 308, Tamil Nadu, India
Natchimuthu Karmegam
Department of Botany, Government Arts College, Salem 636 007, Tamil Nadu, India