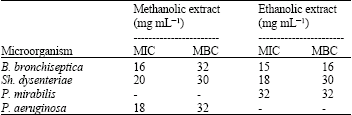Research Article
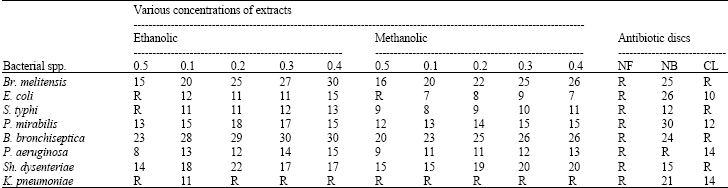
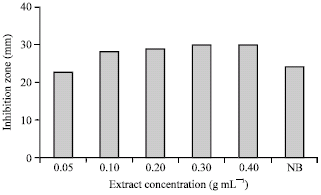
A Preliminary Study on the Antibacterial Activity of Quercus brantii Against Bacterial Pathogens, Particularly Enteric Pathogens
Department of Biology, Faculty of Science, Shahid Chamran University, Ahwaz, Iran
H. Motamedi
Department of Biology, Faculty of Science, Shahid Chamran University, Ahwaz, Iran
S. Maleki
Department of Biology, Faculty of Science, Shahid Chamran University, Ahwaz, Iran
S.M. Seyyednejad
Department of Biology, Faculty of Science, Shahid Chamran University, Ahwaz, Iran