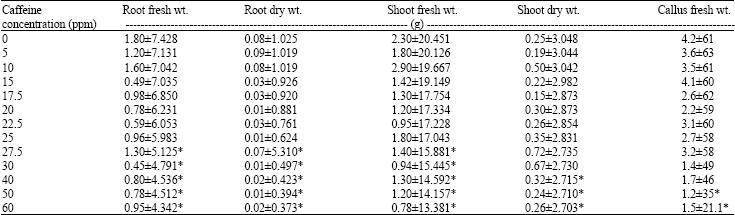
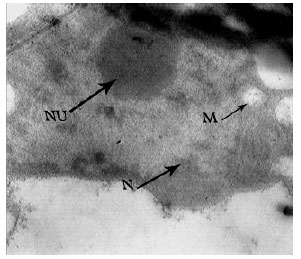
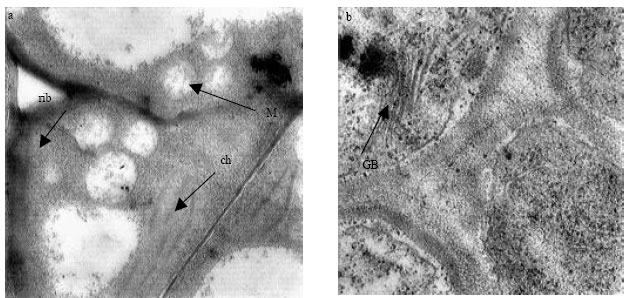
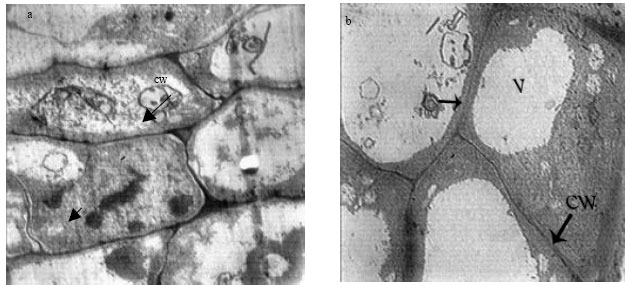
Research Article
Effect of Caffeine on Structure and Ultrastructure of Shoot Apical Meristem of Phaseolus vulgaris L.
Department of Biology, Faculty of Science, Ferdowsi University, Mashad, Iran
Homa Mahmoodzadeh
Department of Biology, Faculty of Science, Islamic Azad University, Mashad, Iran
Samaneh Jamshidi
Department of Biology, Faculty of Science, Ferdowsi University, Mashad, Iran