ABSTRACT
In a previous study, four mutants (M1, M2, M3 and M4) were isolated by exposure of Synechococcus leopoliensis to ultraviolet-B irradiation for 5, 10, 15 and 20 min, respectively. Analyses were performed to make a comparison to the effect of UV on the contents of amino acids, proteins, fatty acids and some enzyme activities of the parent type and mutants. The study shows that total proteins and some amino acids in S. leopoliensis as lysine and arginine decreased by the exposure to UV irradiation, while aspartic increased. Cysteine, alanine and valine completely disappeared from M1, M2 and M3, respectively, while proline disappeared from M1 and M4. A drop in the contents of the aromatic amino acids, phenylalanine and tyrosine occurred in the four mutants. S. leopoliensis showed the disappearance of four fatty acids by the exposure to UV for 10 min, While C17: 1, C 8: 0 and C 15: 0 disappeared from M1, M3 and M4, respectively. C14: 0, which was absent at the parent type, appeared at all the mutants and it was found to increase with increasing the exposure time. All fatty acids of S. leopoliensis, which were present at the parent type, decreased after exposure to UV for 5 min except C15: 0. 10 min and above caused all the fatty acids to decrease with increasing the exposure time except C6: 0, C10: 0, C11: 0, C14: 0, C15: 0 and C18: 3. There was a drop in C18:3 in S. leopoliensis by its exposure to UV for 5 min, which was followed by the increase of that fatty acid with increasing the exposure time. Total saturated, mono, polyunsaturated fatty acids and proteins showed a decrease by increasing the exposure to UV. Enzymes namely μ-esterase, peroxidase and glutamate dehydrogenase were studied for S. leopoliensis and the four mutants. Five minutes exposure to UV increased the activity of peroxidase and glutamate dehydrogenase, but lowered the activity of α-esterase by 93%. More than 5 min exposure to UV caused glutamate dehydrogenase activity to decrease with increasing the exposure time, while peroxidase activity increased. The results of decreased UV-absorbers amino acids in the four mutants suggests that the mutants have a defect in the ability to synthesize them to protect the mutants against the damaging affect of UV radiation. The findings also suggests that the increase of exposure to UV-B irradiance not only affect the contents of amino acids, fatty acids and proteins but also alters enzyme activities in S. leopoliensis.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijb.2007.109.113
URL: https://scialert.net/abstract/?doi=ijb.2007.109.113
INTRODUCTION
The ozone layer shields the earth from harmful UV radiation which has shown to adversely affect a number of photochemical and photobiological processes in a wide variety of aquatic organisms. The effects of ultraviolet irradiation on algae have become an important issue as a man-made depletion of the protective ozone layer has been reported. Enhanced ultraviolet irradiation is considered one of the global environmental problems.
There have been many reports concerning the effects of the ultraviolet irradiation to the growth (Xue et al., 2005), photosynthesis (Huovinen et al., 2006), morphology (Wu et al., 2005), ultrastructure (Holzinger and Lutz, 2006), respiration (Kashian et al., 2004) and enzyme activities (Shiu and Lee, 2005) of algae.
UV irradiation was also found to increase the levels of DNA damage (De Bakker et al., 2005 and Roleda et al., 2006) and change the contents of sugars (Bhandari and Sharma, 2006), pigments (Kim and Lee, 2005), proteins, amino acids (Korbee et al., 2005) and fatty acids (Meireles et al., 2003) of algae.
Analyses were conducted to assess the impact of UV-B doses upon amino acids, proteins, fatty acids contents and some enzyme activities of Synechococcus leopoliensis.
MATERIALS AND METHODS
Strain and culturing: The parent strain Synechococcus leopoliensis Utex 625 was obtained from Utex algal collection, Austen University, USA. Axenic cultures of S. leopolienses were conducted in a culture cabinet and light intensity was 3000 Lux. Culture were grown in GB-11 medium (Vonshak, 1986) under a regime of 16 h light 8 h dark cycles at 35°C and pH 8.
UV-B Irradiation process: Four mutants (M1, M2, M3 and M4) were previously isolated from S. leopoliensis by Noaman et al. (2005) by its exposure to UV for 5, 10, 15 and 20 min, respectively. UV lamp was of a low pressure germicidal one (Uvp, Inc. UVGL-25 San Gabriel, CA, USA).
Amino acids determination: Amino acids determination was performed according to the method of Winder and Eggom (1966). The System used for the analysis was high performance, amino acid analyzer (Sykam, GMBH amino acid analyzer, Germany).
Protein determination: Total proteins were determined according to Hartee (1972).
Fatty acids determination: It was performed according to Radwan (1978).
Enzyme activities estimation: Esterase assay was performed according to Ayene et al. (1988), peroxidase according to Smith et al. (1990), while glutamate dehydrogenase activity was measured as described by Kieronczyk et al. (2003).
RESULTS
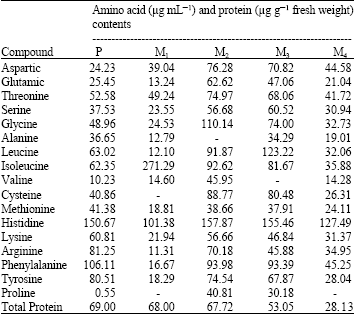
Table 1 shows the contents of amino acids of the parent type Synechococcus leopoliensis and its mutants (M1, M2, M3 and M4) which were previously isolated by Noaman et al. (2005) by exposure of S. leopoliensis to UV-B irradiation for 5, 10, 15 and 20 min, respectively.
Some amino acids in S. leopoliensis as lysine and arginine decreased by the exposure to UV irradiation, while aspartic increased. It must be mentioned that cysteine, alanine and valine completely disappeared from M1, M2 and M3, respectively, while proline disappeared from M1 and M4. A drop in the contents of the aromatic amino acids phenylalanine and tyrosine was noticed in the four mutants especially in M1 and M4. Total proteins decreased by increasing the exposure time of S. leopoliensis to UV irradiation (Table 1).
S. leopoliensis showed the disappearance of four fatty acids by the exposure to UV for 10 min, while C 17: 1, C 8: 0 and C 15: 0 disappeared from M1, M3 and M4, respectively. C 14: 0, which was absent at the parent type, appeared at all the mutants and it was found to be increase with increasing the exposure time to UV (Table 2).
| Table 1: | Amino acids contents of the parent type (P) Synechococcus leopoliensis grown for 21 days and its mutants M1, M2, M3 and produced by exposure to UV-B irradiation |
 | |
| Table 2: | Fatty acids contents of the parent type (P) Synechococcus leopoliensis grown for 21 days and its mutants produced by exposure to UV-B irradiation |
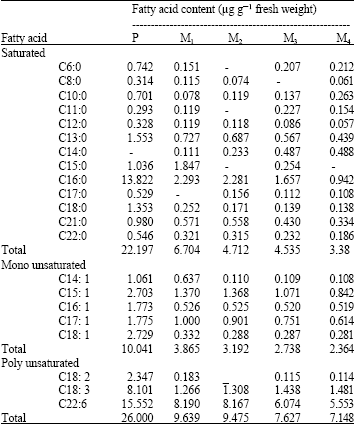 | |
All fatty acids of S. leopoliensis, which were present at the parent type, decreased after exposure to UV for 5 min except C 15: 0. 10 min and above caused all the fatty acids to decrease with increasing the exposure time except C6: 0, C10: 0, C11: 0, C14: 0, C15: 0 and C18: 3. There was a drop in C18:3 in S. leopoliensis by its exposure to UV for 5 min, which was followed by the increase of that fatty acid with increasing the exposure time.
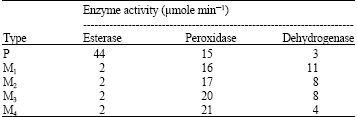
| Table 3: | Enzyme activity of the parent type (P) Synechococcus leopoliensis grown for 21 days and its mutants M1, M2, M3 and M4 produced by exposure to UV-B irradiation |
 | |
Polyunsaturated fatty acid of 22 carbon atoms decreased by increasing the exposure time. Total saturated, mono and polyunsaturated fatty acids of S. leopoliensis showed a decrease by increasing the exposure to UV.
Table 3 showed that 5 min exposure to UV was enough to produce stress to S. leopoliensis causing esterase activity to be lowered by 93%. Activity of peroxidase enzyme increased with increasing the exposure time of S. leopoliensis to UV irradiation, while glutamate dehydrogenase activity decreased with increasing the exposure time except after the first 5 min, which increased the activity nearly four times.
DISCUSSION
Plants produce secondary metabolites that absorb UV and prevent it from penetrating. UV causes oxidative stress as a result of secondary free radical formation (Foyer et al., 1994). Cellular UV absorbers as aromatic amino acids can be activated by UV irradiation and react with molecular oxygen and superoxide radicals (Peak and Peak, 1987).
UV- B radiation decreased or increased the contents of UV- absorbing compounds in many algae (Xue et al., 2005). Wu et al. (2005) observed that there was no significant change in UV-absorbing compounds by exposure of the cyanophyte Arthrospira platensis, suggesting that these compounds were not effectively used as protection against UV radiation and Xuo et al. (2005) stated that the measurements of UV-B absorbing compounds did not necessarily provide a good indicator of tolerance to UV-B.
The increase of some amino acids and decrease of others in S. leopoliensis by exposure to UV-B irradiation were noticed. Alanine decreased at all mutants, a phenomenon which occurred at Phaeocystis pouchetii by its exposure to UV-B irradiation, which was discussed by the damaging effect on the uptake of inorganic nitrogen and nitrogen metabolism (Dohler, 1992).
Aspartic acid in S. leopoliensis, increased by exposure to UV-B irradiation in contrast to Dohler (1984), who found that aspartic acid decreased in Lauderia annulata as a response to UV-B irradiation which was discussed in relation to the impact of UV-B upon carbon and nitrogen metabolism.
Proline showed very high increase in M2 and M3, while it disappeared in M1 and M4. i.e., they lossed the ability to synthesize proline. A three-fold increase in proline occurred in Chlamydomonas nivalis by exposure to UV (Duval et al., 1999), which was accounted by stimulation of UV to the biochemical pathways related to proline metabolism. M2 and M3 were found to accumulate proline, phenomenon that was detected for the first time by Saradhi et al. (1995) who proved that UV-radiation accumulate proline that can protect plant cells against UV-radiation induced peroxidative processes. Although phenylalanine and tyrosine are aromatic amino acids, which can absorb UV-B irradiation (Martin et al., 1985), a drop in their contents in the four mutants was noticed i.e., the mutants were found to have a defect in the ability to synthesize UV-B absorbing amino acids.
S. leopoliensis showed the decrease of total proteins by increasing the exposure time. Damage and degradation of protein by UV is proved in algae (Xue et al., 2005). Kumar et al. (2003) proved the inhibition of nitrogenase enzyme by UV which may be the cause for inhibition of protein synthesis in S. leopoliensis or the damage may be due to the ability of protein to absorb UV which was proved by Ziska and Teramura (1992).
In contrast to Bhandari and Sharma (2006) who found that fatty acid profile of Phormidium corium did not show any qualitative changes due to exposure to UV-B irradiation, S. leopoliensis showed the disappearance of four fatty acids by the exposure to UV for 10 min, While C 17:1, C 8:0 and C 15:0 disappeared from M1, M3 and M4, respectively. C 14: 0, which was absent at the parent type, appeared at all the mutants.
The drop in C 18: 3 in S. leopoliensis by its exposure to UV for 5 min was followed by the increase of that fatty acid of 18 carbon atoms with increasing the exposure time, while polyunsaturated fatty acid of 22 carbon atoms decreased by increasing the exposure time. Kobayashi (1998) found an increase in fatty acids of 18 carbon atoms by UV irradiation but that of 20-22 carbon atoms was not affected by the exposure time.
Ultraviolet irradiation increases fatty acids of Chaetoceros simplex (Boutry et al., 1976) and Pavalvo lutheri (Meireles et al., 2003). UV irradiation resulted in an increase of polyunsaturated fatty acids and a reduction of saturated fatty acids in Phaeodactylum tricornutum and Chaetoceros muellrei (Liang et al., 2006). Total saturated, mono and polyunsaturated fatty acids of S. leopoliensis showed a decrease by increasing the exposure time to UV, which can be discussed by the suggestion of Kobayashi (1998) that UV causes the splitting of fatty acids and the finding of He et al. (2002) that photooxidative damage by UV-radiation, including lipid peroxidation was determined in cyanobacteria.
UV radiation was found to reduce the activity of α-esterase enzyme in S. leopoliensis by 93% after 5 min, which proved that UV had more stress effect on esterase enzyme than peroxidase and glutamate dehydrogenase i.e., the most affected enzyme by UV was esterase, which indicated that it was the most sensitive one. α-esterase was proved by Elsalhin (2004) to be the most affected enzyme in Dunaliella bardawil by the environmental stress.
Exposure of some marine macroalgae to UV radiation resulted in no significant change in the activity of peroxidase (Aguilera et al., 2002). UV radiation was found to increase the activity of peroxidase in S. leopoliensis with increasing the exposure time to UV. Peroxidases are mediators of O2 toxicity and were proved to contribute protection of plants from UV radiation stress (Jansen et al., 2001).
S. leopoliensis exposed 10, 15, or 20 min to UV irradiation have lower activities in glutamate dehydrogenase than those exposed for 5 min. UV was found to affect the process of nitrogen assimilation and its key enzymes like glutamate dehydrogenase (Xu and Zhou, 2004), which in turn affect amino acids synthesis, which appeared highly, injured in M4 (highest exposure time to UV) compared to M2 and M3.
REFERENCES
- Aguilera, J., A. Dummermuth and U. Karsten, 2002. Enzymatic defences against photooxidation tress induced by ultraviolet radiation in Arctic marine macroalgae. Polar Biol., 25: 432-441.
Direct Link - Saradhi, P.P., S. AliaArora and K.V.S.K. Prasad, 1995. Proline accumulates in plants exposed to uv radiation and protects them against uv-induced peroxidation. Biochem. Biophys. Res. Commun., 209: 1-5.
CrossRefDirect Link - Bhandari, R. and P.K. Sharma, 2006. Effect of UV-B on photosynthesis, membrane lipids and MAAs in marine Cyanobacterium Phormidium corium (Agardh) Gomont. Indian. J. Exp. Biol., 44: 330-335.
Direct Link - Boutry, J.L., M. Barbier and M. Ricard, 1976. The marine diatom Chaetoceros simplex calcitrans Paulsen and its enviroment. Effects of light and Ultraviolet Irradiation on the biosynthesis of fatty acids. CR Acad. Sci. D., 282: 239-242.
PubMedDirect Link - De-Bakker, N.V., P.M. van Bodegom and W.H. Von-de-Poll, 2005. Is UV-B radiation affecting charophycean algae in shallow fresh water systems? New Phytol., 166: 957-966.
PubMedDirect Link - Dohler, G., 1992. Impact of UV-B radiation on uptake of 15N-ammonia and 15N-nitrate by phytoplankton of the Wadden Sea. Mar. Biol., 112: 485-489.
Direct Link - Duval, B., K. Shetty and W. Thomas, 1999. Phenolic compounds and antioxidants properties in the snow alga Chlamydomonas nivalis after exposure to UV light. J. Applied Phycol., 11: 559-566.
Direct Link - Foyer, C.H., K. Lelandais and K.J. Kunert, 1994. Photooxidative stress in plants. Physiol. Plant., 92: 696-717.
CrossRef - He, Y.Y., M. Klisch and D.P. Hader, 2002. Adaptation of cyanobacteria to UV-B stress correlated with oxidative stress and oxidative damage. Photochem. Photobiol., 76: 188-196.
Direct Link - Holzinger, A. and C. Lutz, 2006. Algae and UV irradiation: Effects on ultrastructure and related metabolic functions. Micron, 37: 109-207.
Direct Link - Huovinen, P., I. Gomez and C. Lovengreen, 2006. A five year study of solar ultraviolet radiation in Southern Chile (39°S): Potential impact on physiology of costal marine algae. Photochem. Photobiol., 82: 515-522.
Direct Link - Jansen, M.A.K., R.E. Noort, M.Y.A. Lagrimini, M.Y Tan, E. Prinsen, L.M. Lagrimini and R.N. Thorneley, 2001. Phenol-oxidizing peroxidases contribute protection of plants from ultraviolet radiation. Plant Physiol., 26: 1012-1023.
Direct Link - Kashian, D.R., B.A. Prusha and W.H. Clemenrs, 2004. Influence of total organic carbon and UV-B radiation on zinc toxicity and bioaccumulation in aquatic communities. Environ. Sci. Technol., 38: 6371-6376.
Direct Link - Kieronczyk, A., S. Skeie, T. Langsrud and M. Yvon, 2003. Cooperation between Lactococcus lactis and non-starter lactobacilli in the formation of cheese aroma from amino acids. Applied Environ. Microbiol., 61: 734-739.
Direct Link - Kim, S.C. and D.K. Lee, 2005. Inactivation of algal blooms in eutrophic water of drinking water supplies with the photocatalysis of TiO2 thin film on hollow glass beads. Water Sci. Technol., 52: 145-152.
PubMedDirect Link - Kobayashi, Y., 1998. Change of hydroperoxy fatty acids formed by ultraviolet irradiation of bovien retinas-determination of chemiluminescence assay. Nippon Ganka Gakkai Zassi, 102: 15-21.
Direct Link - Korbee, N., F.I. Figueroa and J. Aguilera, 2005. Effect of light quality on the accumulation of photosynthetic pigments, proteins and mycosporine-like amino acids in the red Porphyra leucosticta (Bangiales, Rhodophyta). J. Photochem. Photobiol. B, 80: 71-78.
Direct Link - Kumar, A., M.B. Tyagi, P.N. Jha, G. Srinivas and A. Singh, 2003. Inactivation of cyanobacterial nitrogenase after exposure to ultraviolet-B radiation. Curr. Microbiol., 46: 380-384.
PubMed - Liang, Y., J. Beardall and P. Heaud, 2006. Effects of nitrogen source and UV radiation on the growth, chlorophyll fluorescence and fatty acid composition of Phaeodactylum tricornutum and Chaetoceros muelleri (Bacillariophyceae). J. Photochem. Pholobiol. B, 82: 161-172.
Direct Link - Meireles, L.A., A.C. Guedes and F.X. Malcata, 2003. Increase of yields of eicosapentaenoic and docosahexaenoic acids by the microalga Pavalvo lutheri following random mutagensis. Biotechnol. Bioeng., 81: 50-55.
Direct Link - Radwan, S.S., 1978. Coupling of two-dimensional thin-layer chromatography with gas chromatography for the quantitative analysis of lipid classes and their constituent fatty acids. J. Chromatogr. Sci., 11: 538-542.
CrossRefDirect Link - Roleda, M.Y., C. Wiencke and D. Hanelt, 2006. Thallus morphology and optical Characteristics affect growth and DNA damage by radiation in Juvenile Arctic lamenaria sporophytes. Planta, 223: 407-417.
Direct Link - Shiu, C.T. and T.M. Lee, 2005. Ultraviolet-B-induced oxidative stress and responses of the ascorbate-glutathione cycle in the marine macroalga Ulva fasciata. J. Exp., 56: 2851-2865.
Direct Link - Smith, A.T., N. Santama, S. Dacey, M. Edwards, R.C. Bray, R.N. Thorneley and J.F. Burke, 1990. Expression of a synthetic gene for horse radish peroxidase C in Escherichia coli and folding and activation of the recombinant enzyme with Ca2+ and heme. J. Biol. Chem., 261: 13335-13343.
Direct Link - Xu, Z. and G. Zhou, 2004. Research advance in nitrogen metabolism of plant and its environmental regulation. Ying Yong Sheng Tai Xue Bao, 1: 511-516.
Direct Link - Xue, L., Y. Zhang, T. Zhang, L. An and X. Wang, 2005. Effects of enhanced ultraviolet-B radiation on algae and cyanobacteria. Crit. Rev. Microbiol., 31: 79-89.
Direct Link - Xuo, Y., Q. Liu, B. Lin and H. He, 2005. Physiological responses of 2 year-old Acer davidil seedlings to short-term enhanced UV-B radiation. Ying Yong Sheng Tai Xue Bao, 1: 1682-1986.
Direct Link - Ziska, L.H. and A.H. Teramura, 1992. CO2 enhancement of growth and photosynthesis in Rice (Oryza sativa): Modification by increased ultraviolet-B radiation. Plant Physiol., 99: 473-481.
Direct Link








