Research Article
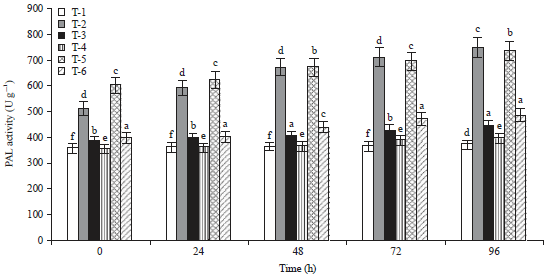
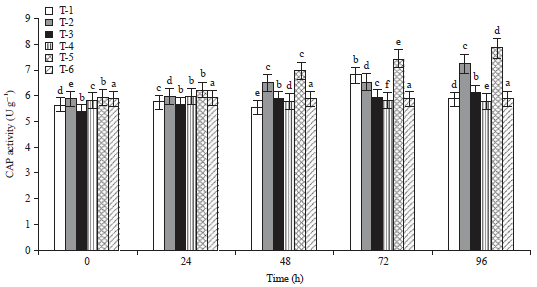
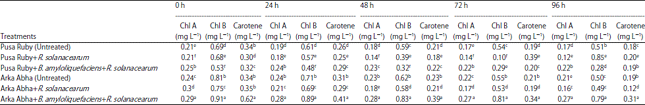
Induction of Defense-related Enzymes by Bacillus amyloliquefaciens DSBA-11 in Resistant and Susceptible Cultivars of Tomato Against Bacterial Wilt Disease
Division of Plant Pathology, Indian Agricultural Research Institute, 110012 New Delhi, India
Dinesh Singh
Division of Plant Pathology, Indian Agricultural Research Institute, 110012 New Delhi, India
LiveDNA: 91.17539
Narendra Kumar
Amity Institutes of Biotechnology, Amity University Haryana, Manesar, 122413 Gurgaon, India
LiveDNA: 91.2651