Research Article
Integrated Management of Chickpea Wilt Incited by Fusarium oxysporum f. sp. ciceris
Department of Plant Protection, 2Department of Genetics and Plant Breeding, Sam Higginbottom Institute of Agriculture Technology and Sciences, Allahabad-211007 (UP), India
S. Zacharia
Department of Plant Protection, 2Department of Genetics and Plant Breeding, Sam Higginbottom Institute of Agriculture Technology and Sciences, Allahabad-211007 (UP), India
K.K. Jaiswal
Department of Plant Protection, 2Department of Genetics and Plant Breeding, Sam Higginbottom Institute of Agriculture Technology and Sciences, Allahabad-211007 (UP), India
P. Pandey
Department of Plant Protection, 2Department of Genetics and Plant Breeding, Sam Higginbottom Institute of Agriculture Technology and Sciences, Allahabad-211007 (UP), India










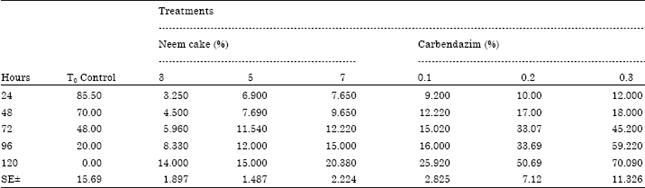
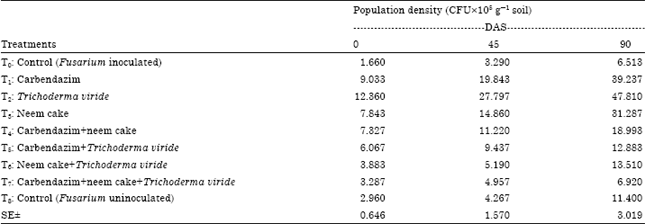
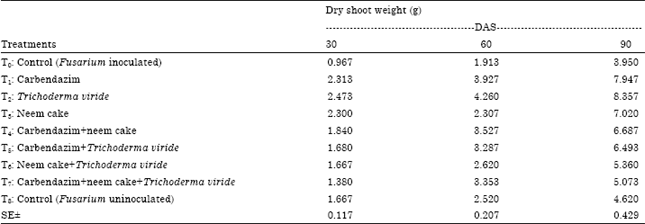
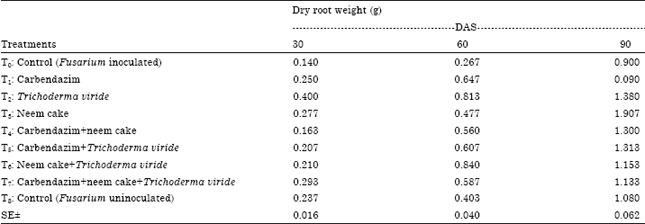

BANGAR DNYANESHWAR KAILAS Reply
I want this paper in Full length