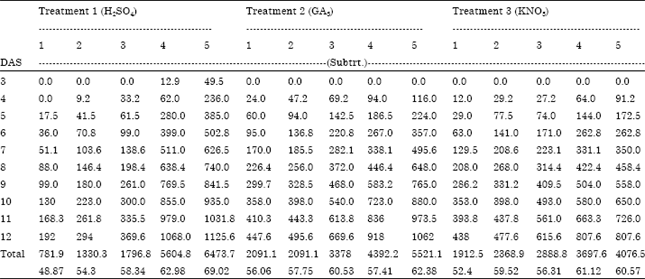
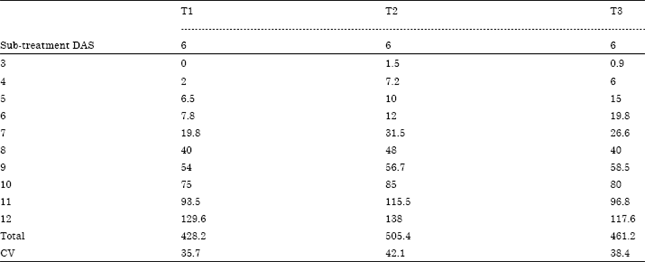
Research Article
Effect of Some Presowing Treatments on Germination of Bauhinia rufescens Seeds
Department of Crop Science, University of Cape Coast, Ghana
G.C. van der Puije
Department of Crop Science, University of Cape Coast, Ghana
K.J. Taah
Department of Crop Science, University of Cape Coast, Ghana
V. Dovlo
Department of Crop Science, University of Cape Coast, Ghana