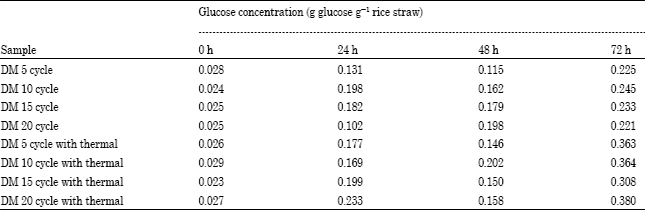
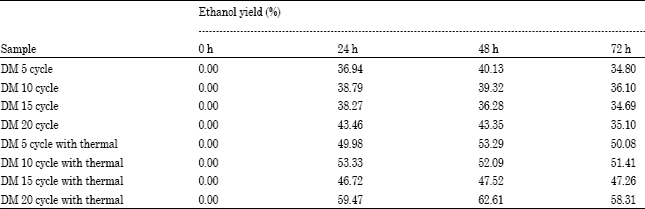
Research Article
Production of Bioethanol from Rice Straw using Cellulase by Local Aspergillus sp.
Bioscience Institute, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
P.L. Yee
Department of Bioprocess Technology,
U.K.M. Shah
Department of Bioprocess Technology,
S.A. Aziz
Department of Bioprocess Technology,
M.A. Hassan
Bioscience Institute, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia