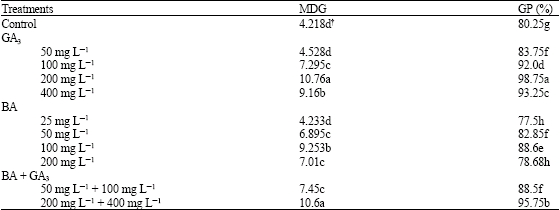
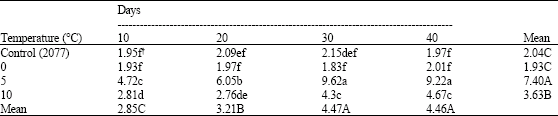
Research Article
Presence of Double Dormancy in Wild Persian Buttercup (Ranunculus asiaticus L.)
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran
M. Khosh-Khui
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran