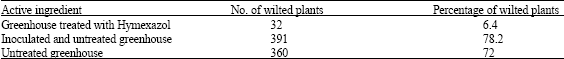Research Article
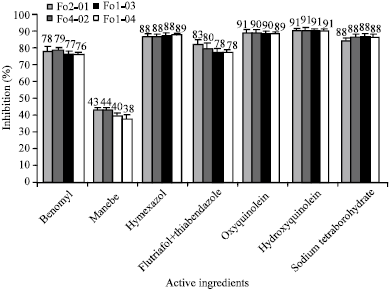
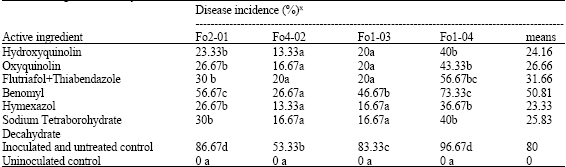
Fusarium Crown and Root Rot of Tomato and its Chemical Control
Department of Phytopathology, Agronomic Higher Institute of Chott-Mariem 4042 Sousse, Tunisia
Mejda Daami-Remadi
National Institute of Agronomic Research, Tunis, PRRDA-CE Chott-Mariem 4042 Sousse, Tunisia
Fakher Ayed
Department of Phytopathology, Agronomic Higher Institute of Chott-Mariem 4042 Sousse, Tunisia
Mohamed El Mahjoub
Department of Phytopathology, Agronomic Higher Institute of Chott-Mariem 4042 Sousse, Tunisia