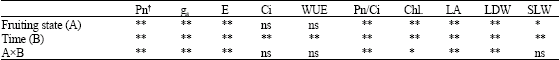
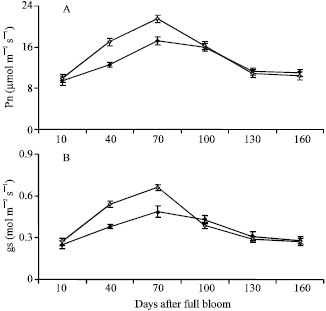
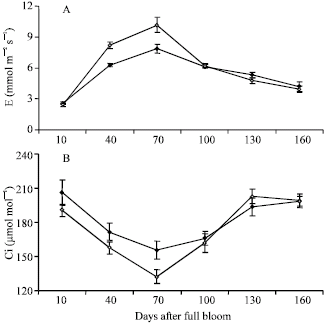
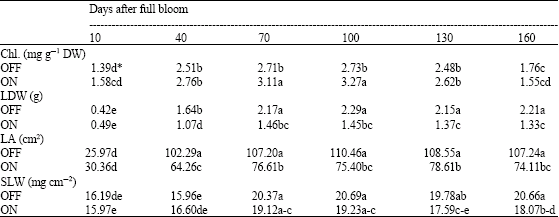
Research Article
Seasonal Variations in Photosynthetic Activities of Pistachio Trees: A Comparison Between Fruiting (ON) and Non-Fruiting (OFF) Trees
Department of Horticulture, College of Agriculture, Shiraz University, Shiraz, Iran
M. Rahemi
Department of Horticulture, College of Agriculture, Shiraz University, Shiraz, Iran
B. Kholdebarin
Department of Biology, College of Science, Shiraz University, Shiraz, Iran