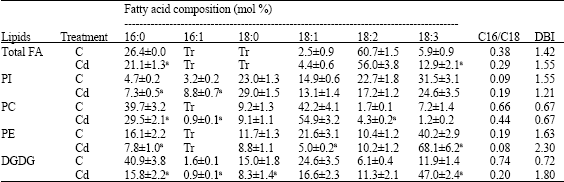
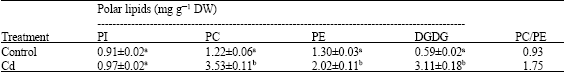
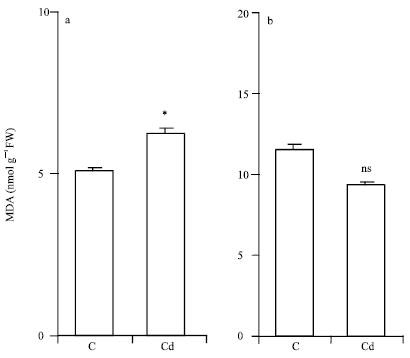
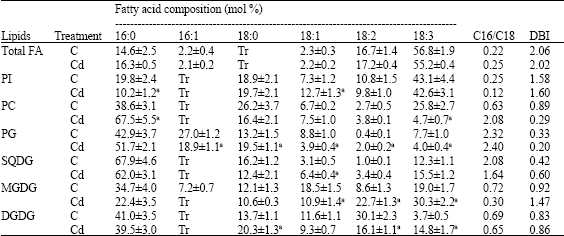
Research Article
Changes in Polar Lipid Composition in Maize Seedlings Induced by Cadmium Stress
Laboratoire de Biologie et Physiologie Cellulaires V6g6tales, Faculte des Sciences de Bizerte, 7021 Zarzmma, Tunisia
Mohamed Anis Elhammadi
Laboratoire de Biologie et Physiologie Cellulaires V6g6tales, Faculte des Sciences de Bizerte, 7021 Zarzmma, Tunisia
Tini Nouhou Seybou
Laboratoire de Biologie et Physiologie Cellulaires V6g6tales, Faculte des Sciences de Bizerte, 7021 Zarzmma, Tunisia
Ali Tekitek
Laboratoire de Biochimie et de Biologie Mol6culaire, Faculte des Sciences de Bizerte, 7021 Zarzmma, Tunisie
Brahim Marzouk
Laboratoire des Ameliorations et Adaptations des Plantes, Institut National de la Recherche Scientifique et Technique (INRST), BP 95-2050 Harnmarn-Lif, Tunisie
Ezzedine El Ferjani
Laboratoire de Biologie et Physiologie Cellulaires V6g6tales, Faculte des Sciences de Bizerte, 7021 Zarzmma, Tunisia