Research Article
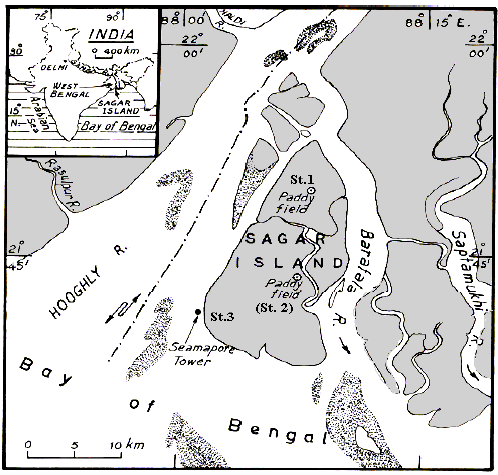
Methane Emission from the Wetland Rice Fields in Sagar Island, Ne Coast of Bay of Bengal, India
Department of Marine Science, Calcutta University, 35, B. C. Road, Calcutta-700019, India
S. K. Mukhopadhayay
Department of Marine Science, Calcutta University, 35, B. C. Road, Calcutta-700019, India
T. K. De
Department of Marine Science, Calcutta University, 35, B. C. Road, Calcutta-700019, India
S. Sen
Department of Chemistry, Calcutta University, 92, A P. C. Road, Calcutta-700009, India
T. K. Jana
Central Salt and Marine Chemical Research Institute, Bhabnagar 364002, Gujrat, India