Research Article
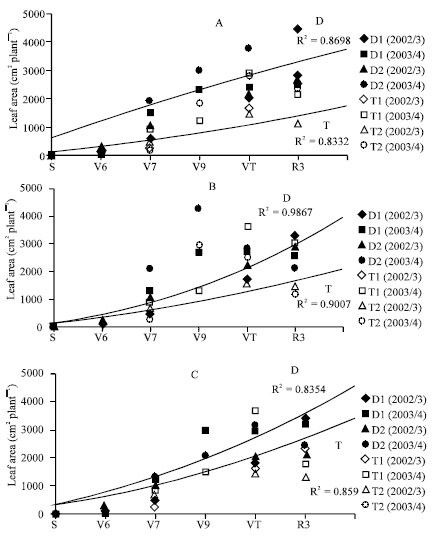
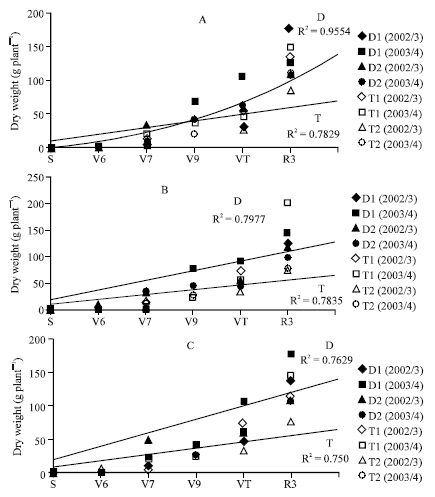
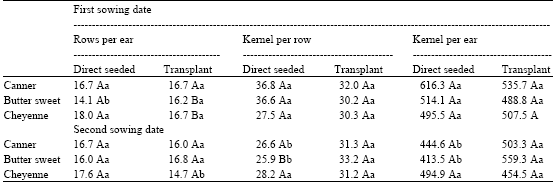
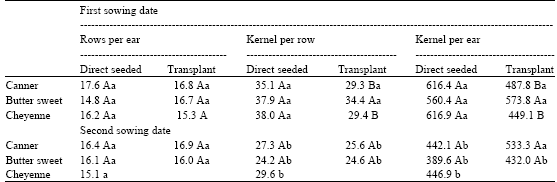
The Effects of Transplant in Sweet Maize (Zea mays L.). I. Growth and Yield
Integrada I.N.T.A. Estaci6n Experimental Agropecuaria Balcarce, Provincia de Buenos Aires, Argentina
Adalberto Di Benedetto
Catedra de Floricultura (FAUBA), Av. San Martin 4453 (1417), Buenos Aires, Argentina
Tomas Gornatti
Integrada I.N.T.A. Estaci6n Experimental Agropecuaria Balcarce, Provincia de Buenos Aires, Argentina