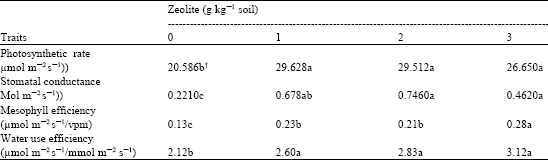
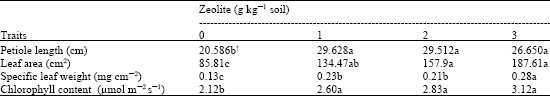
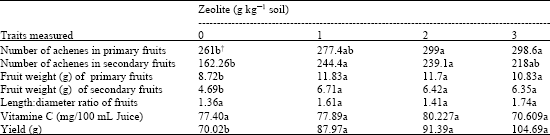
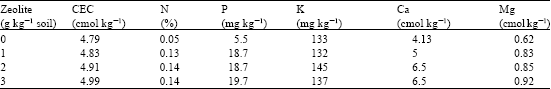Research Article
Effects of Natural Zeolite on Growth and Flowering of Strawberry (Fragariaxananassa Duch.)
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran
M. Khosh-Khui
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran
S. Eshghi
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran