Research Article
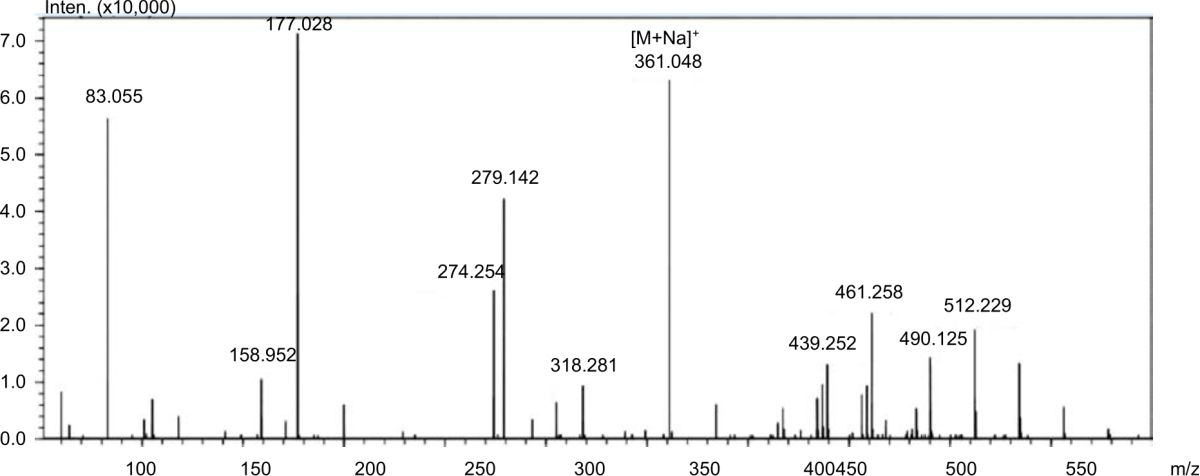
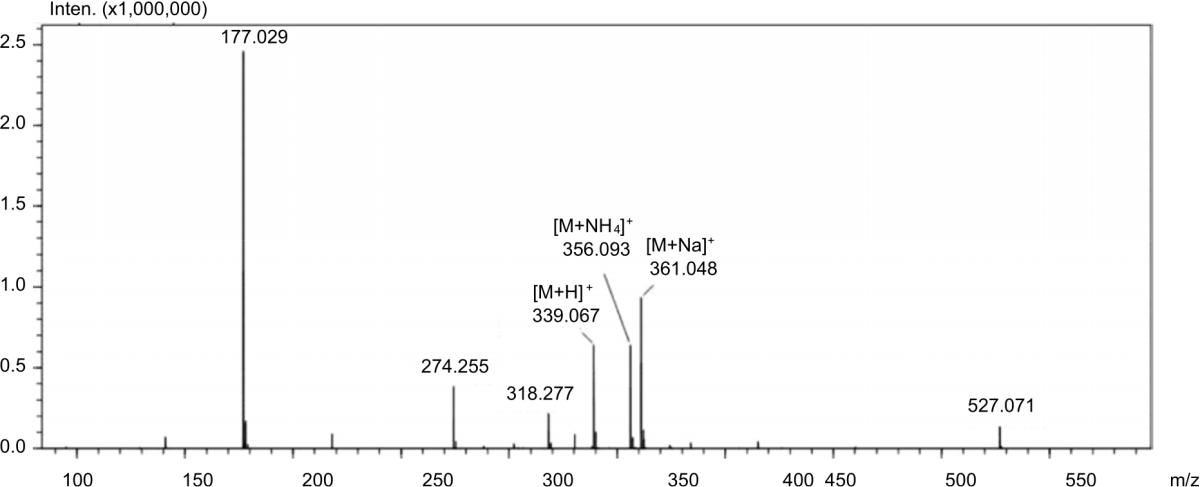
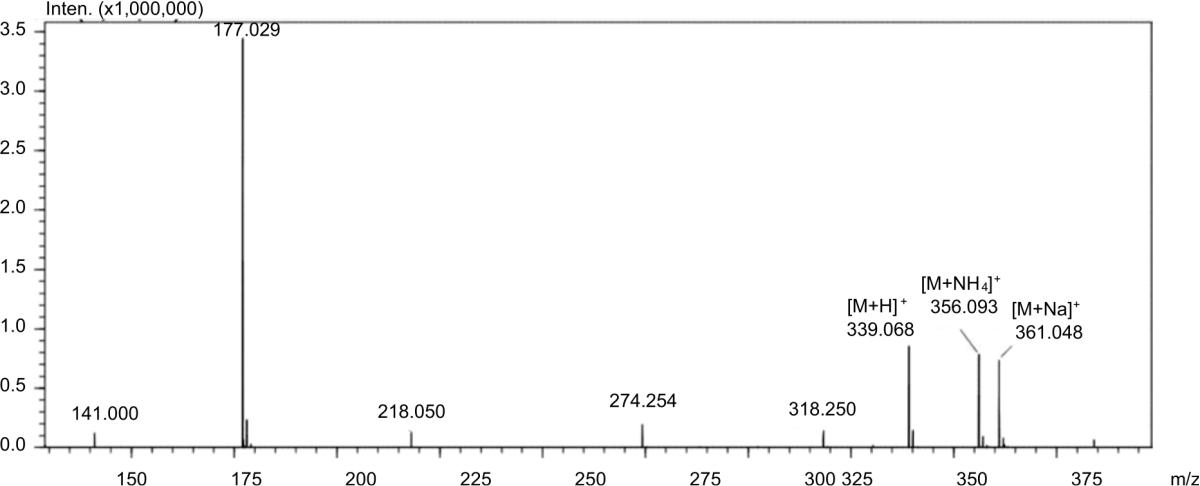
Production Of 2-O-Alpha-D-Glucopyranosyl-L-Ascorbic Acid by Transglycosylation Using Dextransucrase From Leuconostoc mesenteroides
Department of Biochemistry and Microbiology, Rio Claro Biosciences Institute, São Paulo State University, Rio Claro, Av. 24-A, 1515 Bela Vista, Rio Claro, SP, Brazil
Kate Cristina Blanco
São Carlos Institute of Physics, University of São Paulo, Av. Trabalhador São-Carlense, 400, P.O. Box 369, 13560-970 São Carlos, SP, Brazil
LiveDNA: 55.16250
Jonas Contiero
Department of Biochemistry and Microbiology, Rio Claro Biosciences Institute, São Paulo State University, Rio Claro, Av. 24-A, 1515 Bela Vista, Rio Claro, SP, Brazil