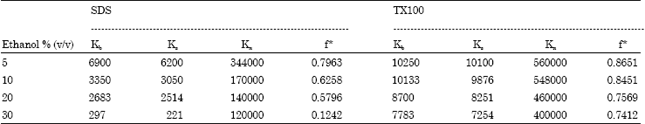
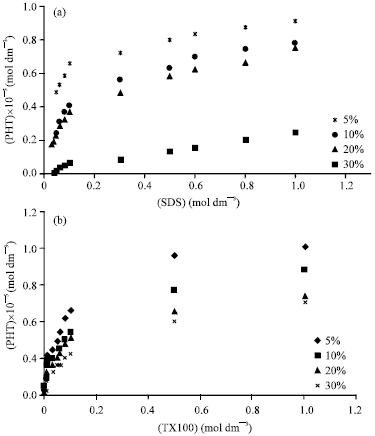
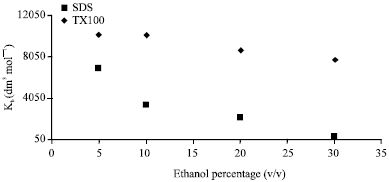
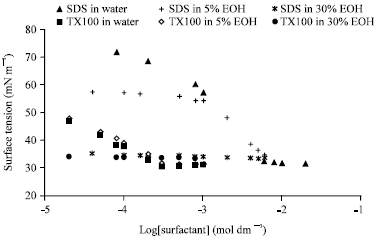
Research Article
Effect of Ethanol on Partition and Binding Equilibrium of Phenothiazine in Anionic and Nonionic Micellar Solutions
General Chemistry Division, Department of Basic Pharmaceutical Sciences, Faculty of Pharmacy, Marmara University, 34718, Uskudar, Istanbul, Turkey
LiveDNA: 90.2658
ORCID: 0000-0001-7979-3020
Umran Var
General Chemistry Division, Department of Basic Pharmaceutical Sciences, Faculty of Pharmacy, Marmara University, 34718, Uskudar, Istanbul, Turkey