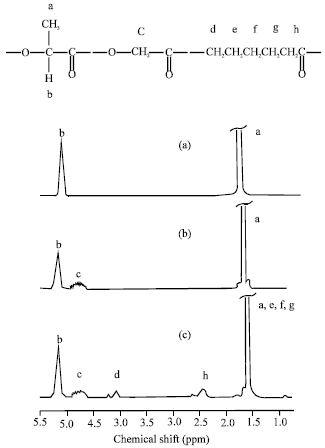
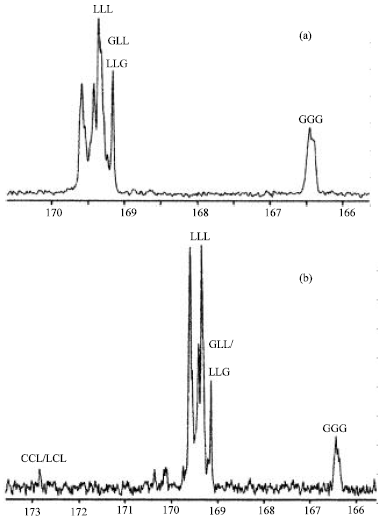
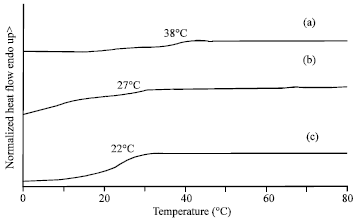
Research Article
Synthesis of Poly (D,L-lactic Acid-co-glycolic Acid-co-ε-caprolactone) Terpolyesters by Direct Polycondensation
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand
LiveDNA: 66.642
M. Srisa-ard
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand
A. Puntumchai
Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand