Research Article
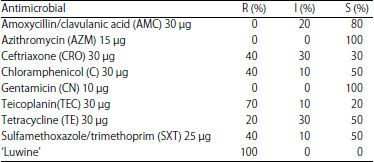
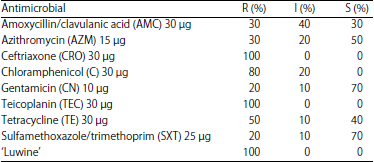
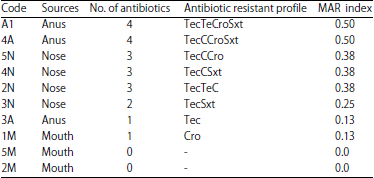
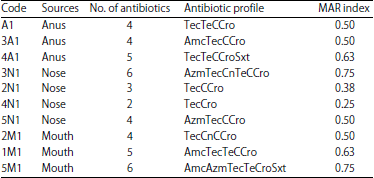
Antibiotic Susceptibility of Fastidious and Non-fastidious Bacteria from African Swine Fever Pigs to Standard Antibiotics and ‘Luwine’
Department of Animal Science, University for Development Studies, P.O. Box TL 1882, Tamale, Ghana
LiveDNA: 233.7256
Godfred Aweligiya
Department of Animal Science, University for Development Studies, P.O. Box TL 1882, Tamale, Ghana
Rejoice Ekli
Department of Animal Science, University for Development Studies, P.O. Box TL 1882, Tamale, Ghana