Short Communication
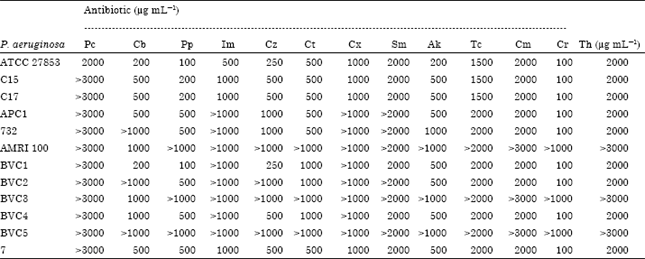
Effective Elimination of Drug Resistance Genes in Pathogenic Pseudomonas aeruginosa by Antipsychotic Agent Thioridazine
Herbicure Healthcare Bio-Herbal Research Foundation, 7 and 8 Metro Garden City, D.H. Road, Pailan, Kolkata-700104, India
S. Chaki
Herbicure Healthcare Bio-Herbal Research Foundation, 7 and 8 Metro Garden City, D.H. Road, Pailan, Kolkata-700104, India
S. Barman
Division of Bacteriology, National Institute of Cholera and Enteric Diseases, P-33, CIT Road, Scheme XM Beliaghata Kolkata-700010, India
S. Das
Department of Physics, Jadavpur University, Kolkata-700032, India
H. Koley
Division of Bacteriology, National Institute of Cholera and Enteric Diseases, P-33, CIT Road, Scheme XM Beliaghata Kolkata-700010, India
S.G. Dastidar
Herbicure Healthcare Bio-Herbal Research Foundation, 7 and 8 Metro Garden City, D.H. Road, Pailan, Kolkata-700104, India