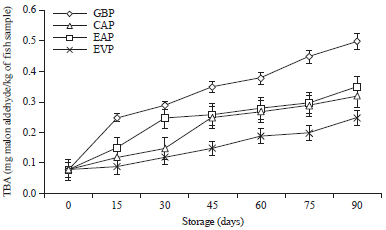
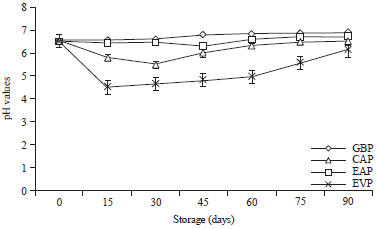
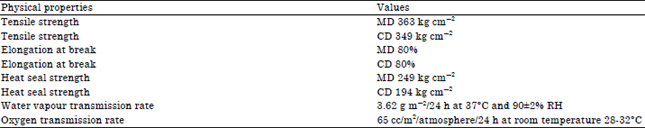
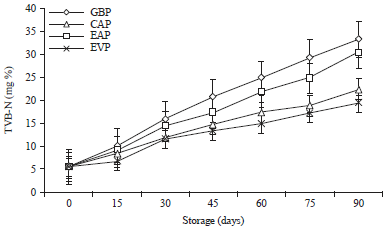
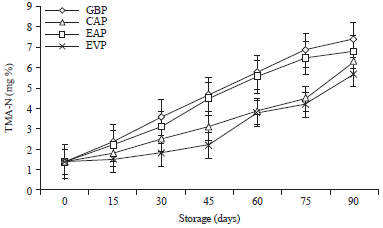
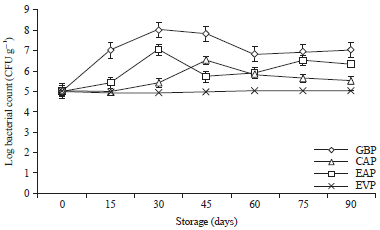
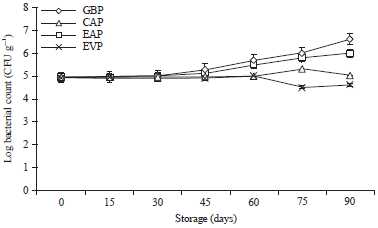
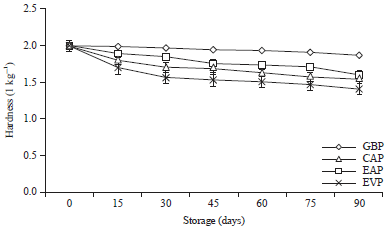
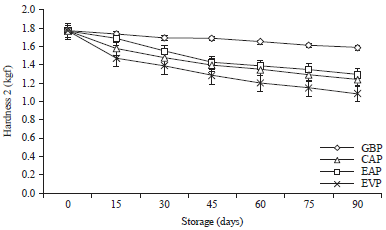
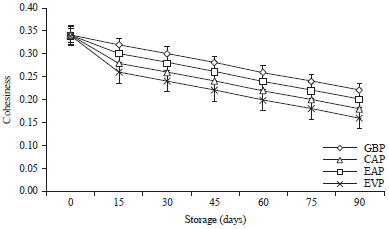
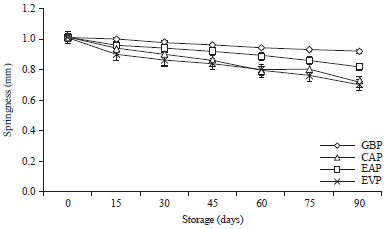
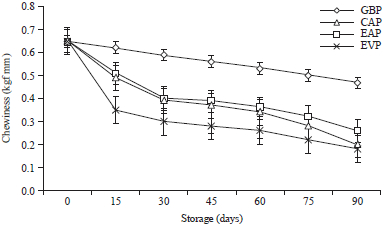
Research Article
Effects of Vacuum-packaging on the Microbiological, Chemical, Textural and Sensory Changes of the Solar Rack Dried Sardines During Chill Storage
Department of Natural Products and Post-Harvest Technology, Marine and Coastal Fisheries Programme, Kenya Marine and Fisheries Research Institute, P.O. Box 81651-80100, Mombasa, Kenya
Odote Peter Michael Oduor
Department of Natural Products and Post-Harvest Technology, Marine and Coastal Fisheries Programme, Kenya Marine and Fisheries Research Institute, P.O. Box 81651-80100, Mombasa, Kenya
Mlanda Mwangemi Nyale
Department of Plant and Microbial Sciences, School of Pure and Applied Sciences, Kenyatta University, P.O. Box 43844-00100, Nairobi, Kenya