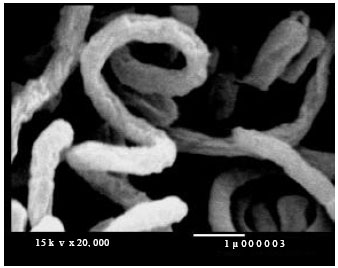
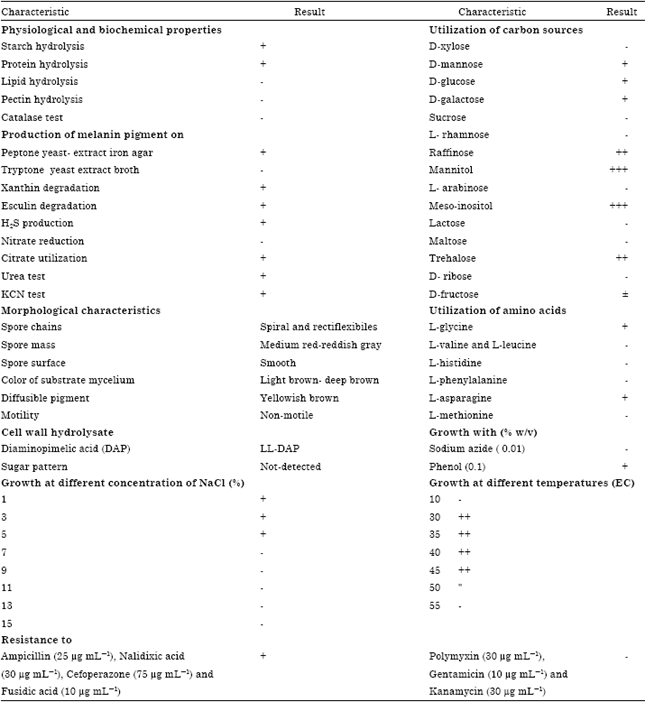
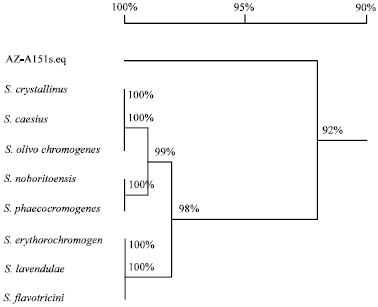
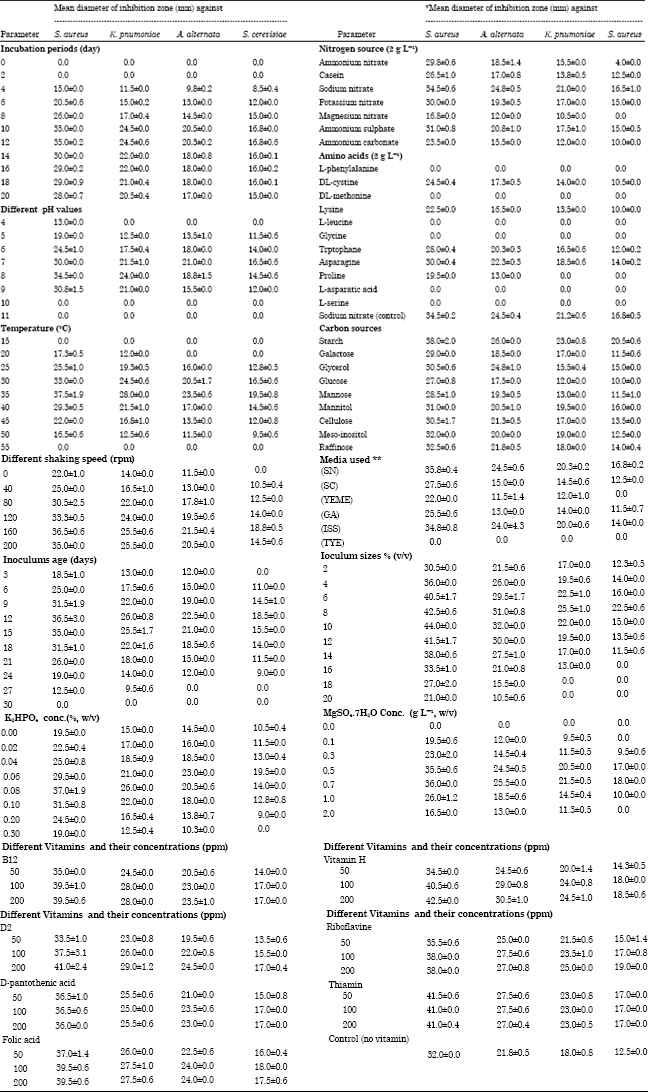
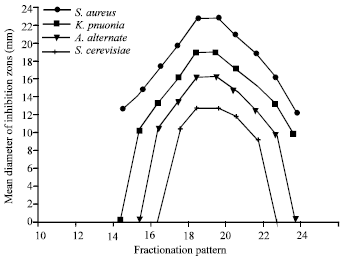
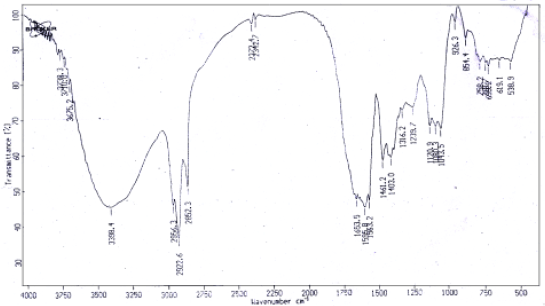
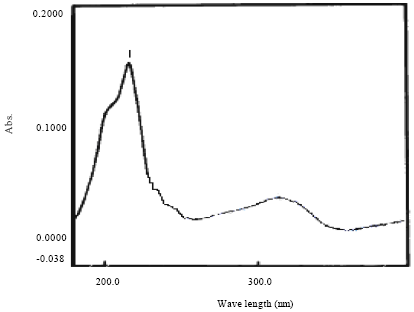
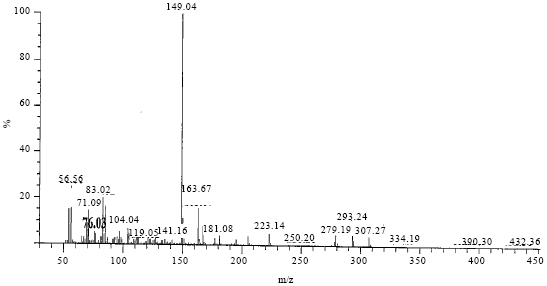
Research Article
Biosynthesis of Hygromycin-B Antibiotic by Streptomyces crystallinus AZ151 Isolated from Assuit, Egypt
Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, Assuit 71524, Egypt
H.M. Atta
Department of Botany and Microbiology, Faculty of Science (Boys), Al-Azhar University, Cairo, Egypt
A.A. Elshanawany
Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, Assuit 71524, Egypt
U.M. Abdoul-raouf
Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, Assuit 71524, Egypt
A.M. El-Adly
Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, Assuit 71524, Egypt