Research Article
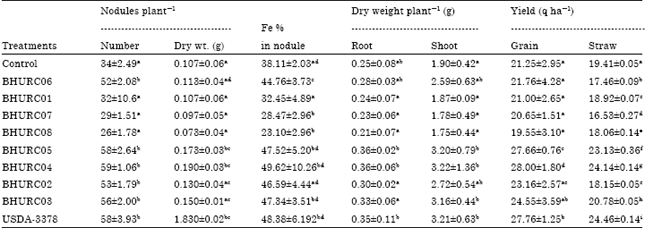
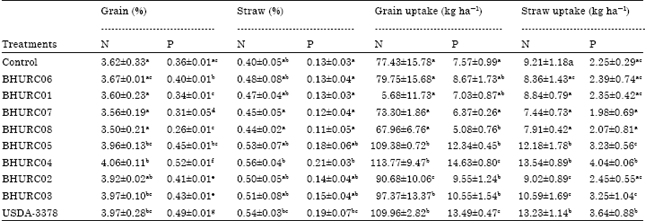
Selection of Effective Indigenous Rhizobium Strain for Seed Inoculation of Chickpea (Cicer aritenium L.) Production
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, UP, India
J.P. Verma
Department of Botany, Faculty of Science, Mahila Mahavidyalaya, Banaras Hindu University, Varanasi-221005, UP, India
LiveDNA: 91.987
ORCID: 0000-0002-2643-9623
V.K. Rajak
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, UP, India
K.N. Tiwari
Department of Botany, Faculty of Science, Mahila Mahavidyalaya, Banaras Hindu University, Varanasi-221005, UP, India