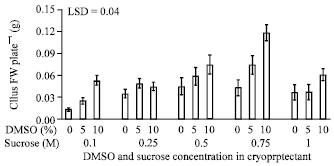
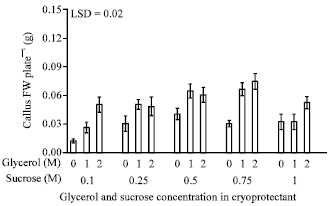
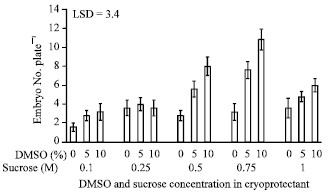
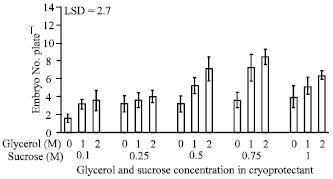
Research Article
Optimizing In vitro Cryopreservation of Date Palm (Phoenix dactylifera L.)
Department of Agricultural Biotechnology, College of Agricultural and Food Sciences, King Faisal University, Al-Hassa 31982, Saudi Arabia
Jameel M. Al-Khayri
Department of Agricultural Biotechnology, College of Agricultural and Food Sciences, King Faisal University, Al-Hassa 31982, Saudi Arabia