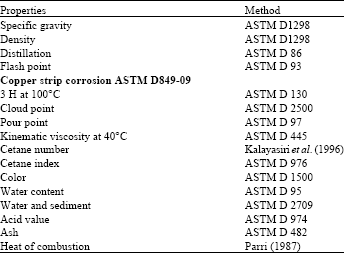
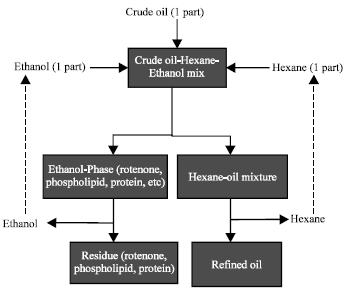
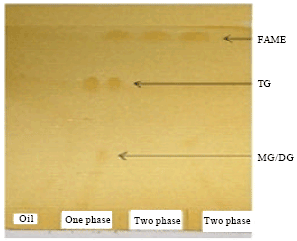
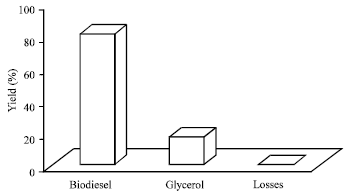
Research Article
Production and Characterization of Biodiesel from Brebra (M. ferruginea) Seed Non-edible Oil
Department of Biotechnology, Faculty of Natural and Computational Sciences, University of Gondar, Ethiopia
Amare Gessesse
Biotechnology Program, Collage of Natural Sciences, Addis Ababa University, Addis Ababa, Ethiopia