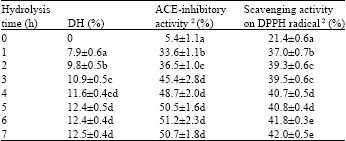
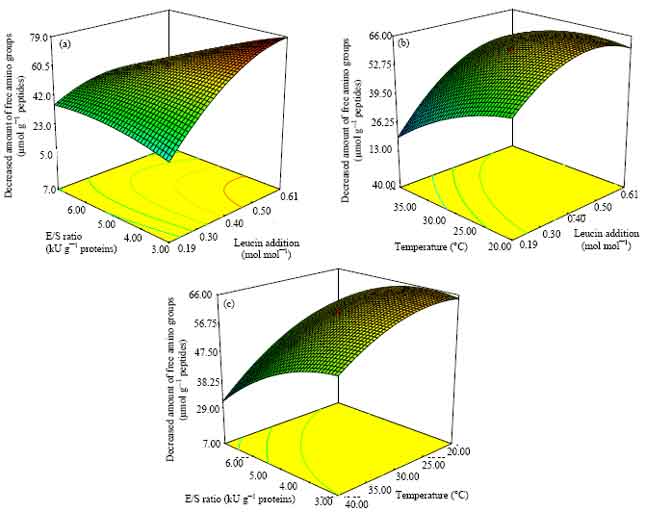
Research Article
In vitro ACE-inhibitory and Antioxidant Activities of the Casein Hydrolysates Subjected to Plastein Reaction with Addition of Three Extrinsic Amino Acids
Key Laboratory of Dairy Science, Ministry of Education, Northeast Agricultural University, Harbin 150030, Peoples� Republic of China
Jing-Ke Wang
Key Laboratory of Dairy Science, Ministry of Education, Northeast Agricultural University, Harbin 150030, Peoples� Republic of China
Tie-Jing Li
Department of Food Science, Northeast Agricultural University, Harbin 150030, Peoples� Republic of China